The Search of a Malaria Vaccine: The Time for Modified Immuno-Potentiating Probes
- PMID: 33540947
- PMCID: PMC7913233
- DOI: 10.3390/vaccines9020115
The Search of a Malaria Vaccine: The Time for Modified Immuno-Potentiating Probes
Abstract
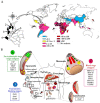
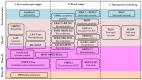
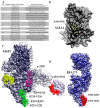
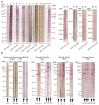
Malaria is a deadly disease that takes the lives of more than 420,000 people a year and is responsible for more than 229 million clinical cases globally. In 2019, 95% of malaria morbidity occurred in African countries. The development of a highly protective vaccine is an urgent task that remains to be solved. Many vaccine candidates have been developed, from the use of the entire attenuated and irradiated pre-erythrocytic parasite forms (or recombinantly expressed antigens thereof) to synthetic candidates formulated in a variety of adjuvants and delivery systems, however these have unfortunately proven a limited efficacy. At present, some vaccine candidates are finishing safety and protective efficacy trials, such as the PfSPZ and the RTS,S/AS01 which are being introduced in Africa. We propose a strategy for introducing non-natural elements into target antigens representing key epitopes of Plasmodium spp. Accordingly, chemical strategies and knowledge of host immunity to Plasmodium spp. have served as the basis. Evidence is obtained after being tested in experimental rodent models for malaria infection and recognized for human sera from malaria-endemic regions. This encourages us to propose such an immune-potentiating strategy to be further considered in the search for new vaccine candidates.
Keywords: Plasmodium spp.; malaria vaccine; non-natural elements; structurally modified antigen.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


References
-
- World Health Organization . World Malaria Report 2020: 20 Years of Global Progress and Challenges. World Health Organization; Geneva, Switzerland: 2020. (electronic version)
-
- Esposito S., Bonanni P., Maggic S., Tand L., Ansaldi F., Lopalco P., Dagan R., Michel J., Damme P., Gaillat J., et al. Recommended immunization schedules for adults: Clinical practice guidelines by the Escmid Vaccine Study Group (EVASG), European Geriatric Medicine Society (EUGMS) and the World Association for Infectious Diseases and Immunological Disorders (WAidid) Hum. Vaccines Immunother. 2016;12:1777–1794. doi: 10.1080/21645515.2016.1150396. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

