A Monovalent and Trivalent MVA-Based Vaccine Completely Protects Mice Against Lethal Venezuelan, Western, and Eastern Equine Encephalitis Virus Aerosol Challenge
- PMID: 33542715
- PMCID: PMC7851092
- DOI: 10.3389/fimmu.2020.598847
A Monovalent and Trivalent MVA-Based Vaccine Completely Protects Mice Against Lethal Venezuelan, Western, and Eastern Equine Encephalitis Virus Aerosol Challenge
Abstract
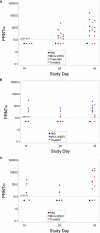
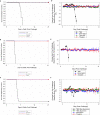
Venezuelan, eastern and western equine encephalitis viruses (EEV) can cause severe disease of the central nervous system in humans, potentially leading to permanent damage or death. Yet, no licensed vaccine for human use is available to protect against these mosquito-borne pathogens, which can be aerosolized and therefore pose a bioterror threat in addition to the risk of natural outbreaks. Using the mouse aerosol challenge model, we evaluated the immunogenicity and efficacy of EEV vaccines that are based on the modified vaccinia Ankara-Bavarian Nordic (MVA-BN®) vaccine platform: three monovalent vaccines expressing the envelope polyproteins E3-E2-6K-E1 of the respective EEV virus, a mixture of these three monovalent EEV vaccines (Triple-Mix) as a first approach to generate a multivalent vaccine, and a true multivalent alphavirus vaccine (MVA-WEV, Trivalent) encoding the polyproteins of all three EEVs in a single non-replicating MVA viral vector. BALB/c mice were vaccinated twice in a four-week interval and samples were assessed for humoral and cellular immunogenicity. Two weeks after the second immunization, animals were exposed to aerosolized EEV. The majority of vaccinated animals exhibited VEEV, WEEV, and EEEV neutralizing antibodies two weeks post-second administration, whereby the average VEEV neutralizing antibodies induced by the monovalent and Trivalent vaccine were significantly higher compared to the Triple-Mix vaccine. The same statistical difference was observed for VEEV E1 specific T cell responses. However, all vaccinated mice developed comparable interferon gamma T cell responses to the VEEV E2 peptide pools. Complete protective efficacy as evaluated by the prevention of mortality and morbidity, lack of clinical signs and viremia, was demonstrated for the respective monovalent MVA-EEV vaccines, the Triple-Mix and the Trivalent single vector vaccine not only in the homologous VEEV Trinidad Donkey challenge model, but also against heterologous VEEV INH-9813, WEEV Fleming, and EEEV V105-00210 inhalational exposures. These EEV vaccines, based on the safe MVA vector platform, therefore represent promising human vaccine candidates. The trivalent MVA-WEV construct, which encodes antigens of all three EEVs in a single vector and can potentially protect against all three encephalitic viruses, is currently being evaluated in a human Phase 1 trial.
Keywords: alphavirus; efficacy; equine encephalitis; modified vaccinia Ankara (MVA); vaccines.
Copyright © 2021 Henning, Endt, Steigerwald, Anderson and Volkmann.
Conflict of interest statement
KE, RS, and AV are employees of Bavarian Nordic GmbH, Germany. RS is an inventor on pending patent applications of Bavarian Nordic A/S, Denmark. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Griffin DE. Alphaviruses. In: Knipe D, Howley PM, editors. Fields Virology, vol. V Lippincott: Williams, and Wilkins; (2007). p. 1023–67.
-
- Tsai TF. Arboviral Infections in the United States. Infect Dis Clin North Am (1991) 5:73–102. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

