A mouse model of stenosis distal to an arteriovenous fistula recapitulates human central venous stenosis
- PMID: 33543148
- PMCID: PMC7857464
- DOI: 10.1016/j.jvssci.2020.07.003
A mouse model of stenosis distal to an arteriovenous fistula recapitulates human central venous stenosis
Abstract
Objective: Central venous stenosis (CVS) is a major cause of arteriovenous fistula (AVF) failure. However, central veins are relatively inaccessible to study with conventional Doppler ultrasound methods. To understand mechanisms underlying AVF failure owing to CVS, an animal model was established that creates a stenosis distal to an AVF. We hypothesized that this mouse model will show comparable morphology and physiology to human CVS.
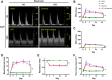
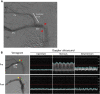
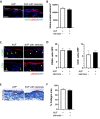
Methods: An aortocaval fistula was created between the distal aorta and inferior vena cava (IVC); a stenosis was then created distal to the fistula by partial IVC ligation. Sham-operated animals, AVF without venous stenosis, and venous stenosis without AVF were used as controls. Physiologic properties of the IVC, both upstream and downstream of the stenosis, or the corresponding sites in models without stenosis, were assessed with ultrasound examination on days 0 to 21. The spectral broadening index was measured to assess the degree of disturbed shear stress. The IVC was harvested at day 21 and specimens were analyzed with immunofluorescence.
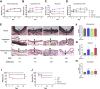
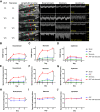
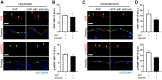
Results: The IVC diameter of mice with an AVF and stenosis showed increased upstream (P = .013), but decreased downstream diameter (P = .001) compared with mice with an AVF but without a stenosis, at all postoperative times (days 3-21). IVC wall thickness increased in mice with an AVF, compared with IVC without an AVF (upstream of stenosis: 13.9 μm vs 11.0 μm vs 4.5 μm vs 3.9 μm; P = .020; downstream of stenosis: 6.0 μm vs 6.6 μm vs μm 4.5 μm vs 3.8 μm; P = .002; AVF with stenosis, AVF, stenosis, sham, respectively). AVF patency significantly decreased in mice with an AVF and stenosis by day 21 (50% vs 90%; P = .048). The IVC of mice with AVF and stenosis showed a venous waveform with pulsatility as well as enhanced velocity at and downstream of the stenosis; similar waveforms were observed in a human case of CVS. Downstream to the stenosis, the spectral broadening index was significantly higher compared with mice with AVF alone (1.06 vs 0.78; P = .011; day 21), and there was a trend towards less immunoreactivity of both Krüppel-like factor 2 and phosphorylated-endothelial nitric oxide synthase compared with mice with an AVF alone.
Conclusions: Partial IVC ligation distal to a mouse aortocaval fistula alters the fistula diameter and wall thickness, decreases patency, and increases distal disturbed flow compared with fistulae without a distal stenosis. Our mouse model of stenosis distal to an AVF may be a faithful representation of human CVS that shows similar morphology and physiology, including disturbed shear stress.
Keywords: Arteriovenous fistula; Central venous stenosis; Disturbed flow; Shear stress; Spectral broadening index.
Conflict of interest statement
Author conflict of interest: none. The editors and reviewers of this article have no relevant financial relationships to disclose per the JVS-Vascular Science policy that requires reviewers to decline review of any manuscript for which they may have a conflict of interest.
Figures


Similar articles
-
Altered hemodynamics during arteriovenous fistula remodeling leads to reduced fistula patency in female mice.JVS Vasc Sci. 2020;1:42-56. doi: 10.1016/j.jvssci.2020.03.001. Epub 2020 Mar 17. JVS Vasc Sci. 2020. PMID: 32754721 Free PMC article.
-
Murine Model of Central Venous Stenosis using Aortocaval Fistula with an Outflow Stenosis.J Vis Exp. 2019 Jul 11;(149). doi: 10.3791/59540. J Vis Exp. 2019. PMID: 31355803
-
Sex hormones impact early maturation and immune response in the arteriovenous fistula mouse model.Am J Physiol Heart Circ Physiol. 2023 Jul 1;325(1):H77-H88. doi: 10.1152/ajpheart.00049.2023. Epub 2023 May 5. Am J Physiol Heart Circ Physiol. 2023. PMID: 37145957 Free PMC article.
-
Disturbed shear stress reduces Klf2 expression in arterial-venous fistulae in vivo.Physiol Rep. 2015 Mar;3(3):e12348. doi: 10.14814/phy2.12348. Physiol Rep. 2015. PMID: 25780089 Free PMC article.
-
[Technique and results of duplex-Doppler for non-stenosing complications of vascular access for chronic hemodialysis: ischemia, steal, high flow rate, aneurysm].J Mal Vasc. 2003 Oct;28(4):200-5. J Mal Vasc. 2003. PMID: 14618110 Review. French.
Cited by
-
Disturbed flow in the juxta-anastomotic area of an arteriovenous fistula correlates with endothelial loss, acute thrombus formation, and neointimal hyperplasia.Am J Physiol Heart Circ Physiol. 2024 Jun 1;326(6):H1446-H1461. doi: 10.1152/ajpheart.00054.2024. Epub 2024 Apr 5. Am J Physiol Heart Circ Physiol. 2024. PMID: 38578237 Free PMC article.
-
A central arteriovenous fistula reduces systemic hypertension in a mouse model.JVS Vasc Sci. 2024 Feb 15;5:100191. doi: 10.1016/j.jvssci.2024.100191. eCollection 2024. JVS Vasc Sci. 2024. PMID: 38510938 Free PMC article.
-
Great start predicts bright future for JVS-Vascular Science.JVS Vasc Sci. 2021 Dec 27;2:287-291. doi: 10.1016/j.jvssci.2021.11.001. eCollection 2021. JVS Vasc Sci. 2021. PMID: 35112087 Free PMC article. No abstract available.
References
-
- Saran R., Robinson B., Abbott K.C., Bragg-Gresham J., Chen X., Gipson D. US Renal Data System 2019 Annual Data Report: epidemiology of kidney disease in the United States. Am J Kidney Dis. 2020;75:A6–A7. - PubMed
-
- Almasri J., Alsawas M., Mainou M., Mustafa R.A., Wang Z., Woo K. Outcomes of vascular access for hemodialysis: a systematic review and meta-analysis. J Vasc Surg. 2016;64:236–243. - PubMed
-
- Murad M.H., Elamin M.B., Sidawy A.N., Malaga G., Rizvi A.Z., Flynn D.N. Autogenous versus prosthetic vascular access for hemodialysis: a systematic review and meta-analysis. J Vasc Surg. 2008;48:34S–47S. - PubMed
-
- Donca I.Z., Wish J.B. Systemic barriers to optimal hemodialysis access. Semin Nephrol. 2012;32:519–529. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

