Preclinical Pharmacokinetic Interaction and Histopathological Analyses of Hedyotis diffusa on Sorafenib in Rats
- PMID: 33553923
- PMCID: PMC7860071
- DOI: 10.1021/acsomega.0c05461
Preclinical Pharmacokinetic Interaction and Histopathological Analyses of Hedyotis diffusa on Sorafenib in Rats
Abstract
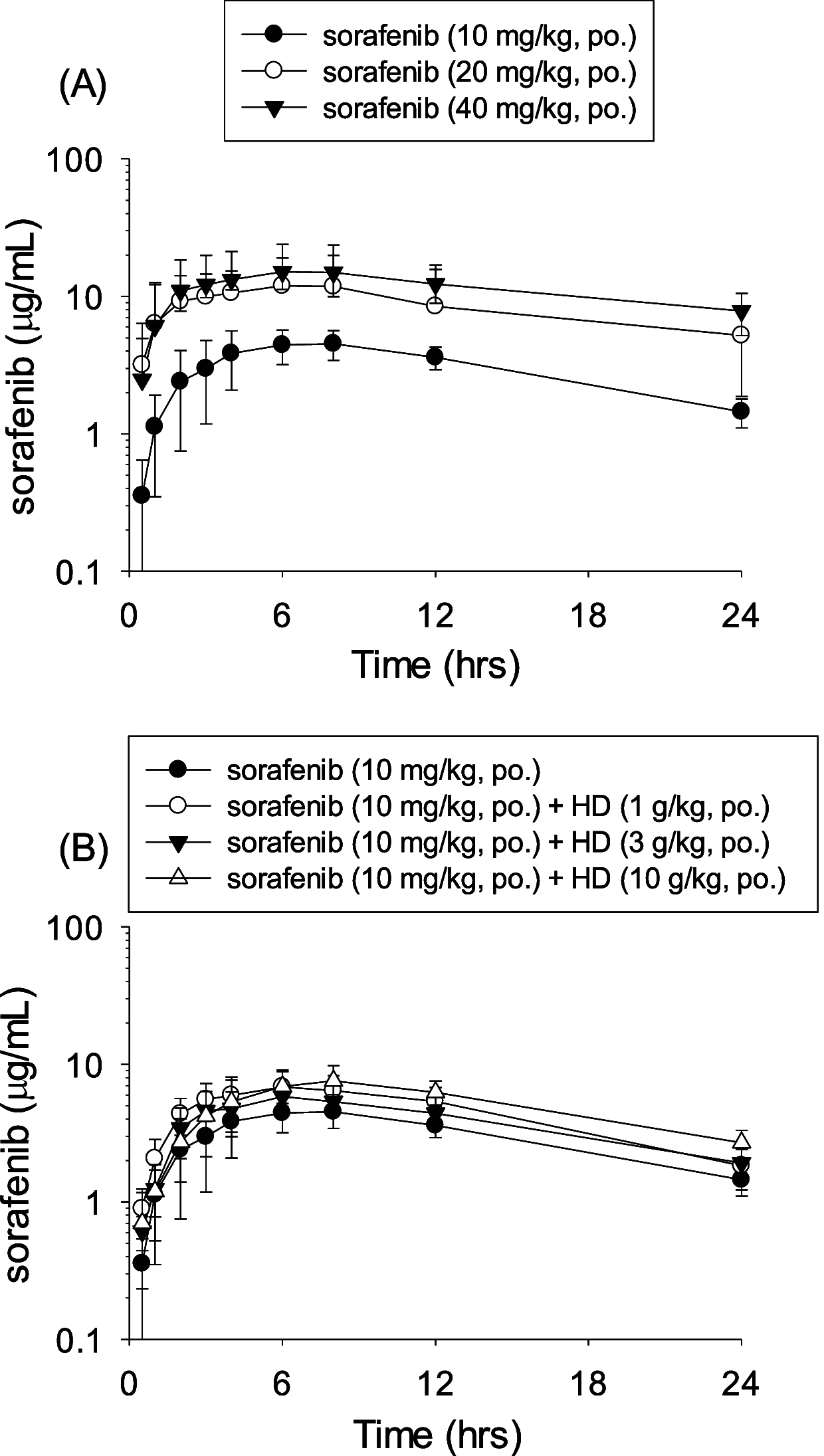
Sorafenib is one of the most effective target therapeutic agents for patients with late-stage hepatocellular carcinoma. To seek possible alternative adjuvant agents to enhance the efficacy and improve the side effect of sorafenib, Hedyotis diffusa, one of the most prescribed phytomedicines for treating liver cancer patients in Taiwan, was evaluated in this work. We hypothesized that H. diffusa extract is a safety herb combination on the pharmacokinetic and pharmacodynamic effects of sorafenib. We designed treatments of sorafenib in combination with or without H. diffusa extract to examine its pharmacokinetic properties and effects on liver inflammation. The HPLC-photodiode-array method was designed for monitoring the plasma level and pharmacokinetic parameter of sorafenib in rat plasma. The pharmacokinetic results demonstrated that the area under the curve of sorafenib (10 mg/kg, p.o.) in combination with various doses of H. diffusa formulation (1, 3, and 10 g/kg, p.o.) for 5 consecutive days were 5560 ± 1392, 7965 ± 2055, 7271 ± 1371, and 8821 ± 1705 min μg/mL, respectively, no significant difference when compared with sorafenib treatment alone. Furthermore, the hepatic activity in rats administered with sorafenib with/without H. diffusa extract was quantitatively scored by modified hepatic activity index grading. H. diffusa extract in the range of 1 to 10 g/kg per day did not elicit significant herb-induced hepatotoxicity in rats, based on the histopathological study. Consequently, our findings provided positive safety outcomes for the administration of sorafenib in combination with the phytomedicine H. diffusa.
© 2021 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
Dose-dependent effects of Hedyotis diffusa extract on the pharmacokinetics of tamoxifen, 4-hydroxytamoxifen, and N-desmethyltamoxifen.Biomed Pharmacother. 2022 Jan;145:112466. doi: 10.1016/j.biopha.2021.112466. Epub 2021 Nov 25. Biomed Pharmacother. 2022. PMID: 34839255
-
[The Component Analysis of Petroleum Ether Extract in Scutellariae barbatae,Hedyotis diffusa and the Herb Pair and the Investigation of Anti-Endometrial Cancer Cells Activity].Zhong Yao Cai. 2016 Apr;39(4):789-94. Zhong Yao Cai. 2016. PMID: 30132589 Chinese.
-
Toxicity study of compound granules of Hedyotis diffusa: Acute toxicity and long-term toxicity.J Ethnopharmacol. 2024 Mar 1;321:117434. doi: 10.1016/j.jep.2023.117434. Epub 2023 Nov 20. J Ethnopharmacol. 2024. PMID: 37992881
-
Clarification of the phenotypic characteristics and anti-tumor activity of Hedyotis diffusa.Am J Chin Med. 2011;39(1):201-13. doi: 10.1142/S0192415X11008750. Am J Chin Med. 2011. PMID: 21213409
-
Chemical and preclinical studies on Hedyotis diffusa with anticancer potential.J Asian Nat Prod Res. 2013;15(5):550-65. doi: 10.1080/10286020.2013.781589. Epub 2013 Apr 22. J Asian Nat Prod Res. 2013. PMID: 23600735 Review.
Cited by
-
Sorafenib plus memory-like natural killer cell immunochemotherapy boosts treatment response in liver cancer.BMC Cancer. 2024 Sep 30;24(1):1215. doi: 10.1186/s12885-024-12718-4. BMC Cancer. 2024. PMID: 39350084 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

