Severity of liver test abnormalities in coronavirus disease 2019 depends on comorbidities and predicts early in-hospital mortality
- PMID: 33560687
- PMCID: PMC8734572
- DOI: 10.1097/MEG.0000000000002055
Severity of liver test abnormalities in coronavirus disease 2019 depends on comorbidities and predicts early in-hospital mortality
Abstract
Background and aims: Liver chemistry abnormalities (LCA) are common in patients with coronavirus disease 2019 (COVID-19), but their causes and clinical impact have not been adequately studied. We assessed the associations between LCA and clinical characteristics, inflammatory serum markers, in-hospital mortality.
Methods: Ten thousand eight hundred fifty-six adult patients with COVID-19 hospitalized in 13 hospitals in New York (1 March to 27 April 2020) were analyzed retrospectively. Abnormalities of liver chemistries [aspartate aminotransferase (AST), alanine aminotransferase, alkaline phosphatase, or total bilirubin] were defined as absent, mild-moderate (at least one value up to four times elevated), or severe.
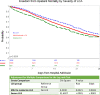
Results: LCA were mild-moderate in 63.9% and severe in 7.6% at admission. Risk factors for severe LCA were male sex and chronic liver disease. Conversely, hypertension and diabetes mellitus were less likely associated with severe LCA. AST elevation correlated weakly to modestly with inflammatory markers. On adjusted analysis, in-hospital mortality was 1.56 times and 1.87 times increased in patients with mild-to-moderate and severe LCA, respectively. Diabetes, hypertension, male sex, and age greater than 60 years was associated with incremental risk of mortality with increase severity of LCA, especially in the first week of hospitalization. HTN was not associated with increased in-hospital mortality unless LCA was present.
Conclusion: Increasing severity of LCA on hospital admission predicts early in-hospital mortality in COVID-19 patients. Mortality associated with the known risk factors, hypertension, diabetes, male sex, and old age was accentuated in the presence of LCA. AST correlated modestly with inflammatory markers.
Copyright © 2020 Wolters Kluwer Health, Inc. All rights reserved.
Conflict of interest statement
Sanjaya K. Satapathy has served as a speaker for Intercept, Alexion, Dova, as an advisory board member for Gilead, Intercept, Bayer and has received research funding from Gilead, Biotest, Genfit, Conatus, Intercept, Shire, Exact Sciences, Eananta, Dova, Bayer. Sanjaya K. Satapathy is an employee of Northwell Health. The remaining authors declare no conflicts of interest.
Figures

References
-
- Qiu H, Wander P, Bernstein D, Satapathy SK. Acute on chronic liver failure from novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Liver Int 2020; 40:1590–1593. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

