HMGB1 regulates ferroptosis through Nrf2 pathway in mesangial cells in response to high glucose
- PMID: 33565572
- PMCID: PMC7897919
- DOI: 10.1042/BSR20202924
HMGB1 regulates ferroptosis through Nrf2 pathway in mesangial cells in response to high glucose
Abstract
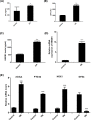
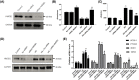
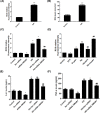
Ferroptosis, a novel type of programmed cell death, is involved in inflammation and oxidation of various human diseases, including diabetic kidney disease. The present study explored the role of high-mobility group box-1 (HMGB1) on the regulation of ferroptosis in mesangial cells in response to high glucose. Compared with healthy control, levels of serum ferritin, lactate dehydrogenase (LDH), reactive oxygen species (ROS), malonaldehyde (MDA), and HMGB1 were significantly elevated in diabetic nephropathy (DN) patients, accompanied with deregulated ferroptosis-related molecules, including long-chain acyl-CoA synthetase 4 (ACSL4), prostaglandin-endoperoxide synthase 2 (PTGS2), NADPH oxidase 1 (NOX1), and glutathione peroxidase 4 (GPX4). In vitro assay revealed that erastin and high glucose both induced ferroptosis in mesangial cells. Suppression of HMGB1 restored cellular proliferation, prevented ROS and LDH generation, decreased ACSL4, PTGS2, and NOX1, and increased GPX4 levels in mesangial cells. Furthermore, nuclear factor E2-related factor 2 (Nrf2) was decreased in DN patients and high glucose-mediated translocation of HMGB1 in mesangial cells. Knockdown of HMGB1 suppressed high glucose-induced activation of TLR4/NF-κB axis and promoted Nrf2 expression as well as its downstream targets including HO-1, NQO-1, GCLC, and GCLM. Collectively, these findings suggest that HMGB1 regulates glucose-induced ferroptosis via Nrf2 pathway in mesangial cells.
© 2021 The Author(s).
Conflict of interest statement
The authors declare that there are no competing interests associated with the manuscript.
Figures
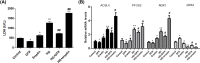
Similar articles
-
circLRP6 regulates high glucose-induced proliferation, oxidative stress, ECM accumulation, and inflammation in mesangial cells.J Cell Physiol. 2019 Nov;234(11):21249-21259. doi: 10.1002/jcp.28730. Epub 2019 May 13. J Cell Physiol. 2019. PMID: 31087368
-
SYVN1 attenuates ferroptosis and alleviates spinal cord ischemia-reperfusion injury in rats by regulating the HMGB1/NRF2/HO-1 axis.Int Immunopharmacol. 2023 Oct;123:110802. doi: 10.1016/j.intimp.2023.110802. Epub 2023 Aug 15. Int Immunopharmacol. 2023. PMID: 37591122
-
Umbelliferone delays the progression of diabetic nephropathy by inhibiting ferroptosis through activation of the Nrf-2/HO-1 pathway.Food Chem Toxicol. 2022 May;163:112892. doi: 10.1016/j.fct.2022.112892. Epub 2022 Mar 10. Food Chem Toxicol. 2022. PMID: 35278496
-
[Roles of ferroptosis in the development of diabetic nephropathy].Zhejiang Da Xue Xue Bao Yi Xue Ban. 2024 Dec 25;53(6):708-714. doi: 10.3724/zdxbyxb-2024-0114. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2024. PMID: 39757741 Free PMC article. Review. Chinese.
-
Concerted action of Nrf2-ARE pathway, MRN complex, HMGB1 and inflammatory cytokines - implication in modification of radiation damage.Redox Biol. 2014 Feb 28;2:832-46. doi: 10.1016/j.redox.2014.02.008. eCollection 2014. Redox Biol. 2014. PMID: 25009785 Free PMC article. Review.
Cited by
-
Translational and Clinical Significance of DAMPs, PAMPs, and PRRs in Trauma-induced Inflammation.Arch Clin Biomed Res. 2022;6(5):673-685. doi: 10.26502/acbr.50170279. Epub 2022 Aug 26. Arch Clin Biomed Res. 2022. PMID: 36147548 Free PMC article.
-
Targeting ferroptosis: a new therapeutic opportunity for kidney diseases.Front Immunol. 2024 Jul 3;15:1435139. doi: 10.3389/fimmu.2024.1435139. eCollection 2024. Front Immunol. 2024. PMID: 39021564 Free PMC article. Review.
-
The Potential Role of Ferroptosis in Systemic Lupus Erythematosus.Front Immunol. 2022 Apr 21;13:855622. doi: 10.3389/fimmu.2022.855622. eCollection 2022. Front Immunol. 2022. PMID: 35529869 Free PMC article. Review.
-
Targeting ferroptosis as a prospective therapeutic approach for diabetic nephropathy.Ann Med. 2024 Dec;56(1):2346543. doi: 10.1080/07853890.2024.2346543. Epub 2024 Apr 24. Ann Med. 2024. PMID: 38657163 Free PMC article. Review.
-
Defective NCOA4-dependent ferroptosis in senescent fibroblasts retards diabetic wound healing.Cell Death Discov. 2023 Apr 28;9(1):138. doi: 10.1038/s41420-023-01437-7. Cell Death Discov. 2023. PMID: 37117222 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

