Pharmacoepidemiological Evaluation in Prostate Cancer-Common Pitfalls and How to Avoid Them
- PMID: 33572236
- PMCID: PMC7914977
- DOI: 10.3390/cancers13040696
Pharmacoepidemiological Evaluation in Prostate Cancer-Common Pitfalls and How to Avoid Them
Abstract
Pharmacoepidemiologic research provides opportunities to evaluate how commonly used drug groups, such as cholesterol-lowering or antidiabetic drugs, may affect the prostate cancer risk or mortality. This type of research is valuable in estimating real-life drug effects. Nonetheless, pharmacoepidemiological studies are prone to multiple sources of bias that mainly arise from systematic differences between medication users and non-users. If these are not appreciated and properly controlled for, there is a risk of obtaining biased results and reaching erroneous conclusions. Therefore, in order to improve the quality of future research, we describe common biases in pharmacoepidemiological studies, particularly in the context of prostate cancer research. We also list common ways to mitigate these biases and to estimate causality between medication use and cancer outcomes.
Keywords: common biases; confounding; metabolism; pharmacoepidemiology; prostate cancer; retrospective studies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

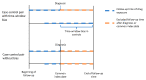

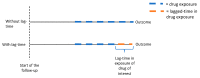
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

