Can Be miR-126-3p a Biomarker of Premature Aging? An Ex Vivo and In Vitro Study in Fabry Disease
- PMID: 33572275
- PMCID: PMC7915347
- DOI: 10.3390/cells10020356
Can Be miR-126-3p a Biomarker of Premature Aging? An Ex Vivo and In Vitro Study in Fabry Disease
Abstract
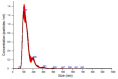
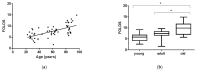
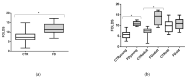
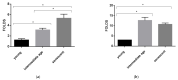
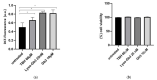
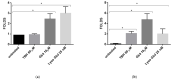
Fabry disease (FD) is a lysosomal storage disorder (LSD) characterized by lysosomal accumulation of glycosphingolipids in a wide variety of cytotypes, including endothelial cells (ECs). FD patients experience a significantly reduced life expectancy compared to the general population; therefore, the association with a premature aging process would be plausible. To assess this hypothesis, miR-126-3p, a senescence-associated microRNA (SA-miRNAs), was considered as an aging biomarker. The levels of miR-126-3p contained in small extracellular vesicles (sEVs), with about 130 nm of diameter, were measured in FD patients and healthy subjects divided into age classes, in vitro, in human umbilical vein endothelial cells (HUVECs) "young" and undergoing replicative senescence, through a quantitative polymerase chain reaction (qPCR) approach. We confirmed that, in vivo, circulating miR-126 levels physiologically increase with age. In vitro, miR-126 augments in HUVECs underwent replicative senescence. We observed that FD patients are characterized by higher miR-126-3p levels in sEVs, compared to age-matched healthy subjects. We also explored, in vitro, the effect on ECs of glycosphingolipids that are typically accumulated in FD patients. We observed that FD storage substances induced in HUVECs premature senescence and increased of miR-126-3p levels. This study reinforces the hypothesis that FD may aggravate the normal aging process.
Keywords: Fabry disease; HUVEC; aging; endothelial cells; miR-126-3p; microRNA; senescence; small extracellular vesicles.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

