Outer Hair Cell Glutamate Signaling through Type II Spiral Ganglion Afferents Activates Neurons in the Cochlear Nucleus in Response to Nondamaging Sounds
- PMID: 33574178
- PMCID: PMC8018895
- DOI: 10.1523/JNEUROSCI.0619-20.2021
Outer Hair Cell Glutamate Signaling through Type II Spiral Ganglion Afferents Activates Neurons in the Cochlear Nucleus in Response to Nondamaging Sounds
Abstract
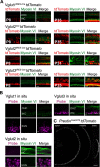
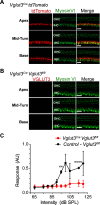
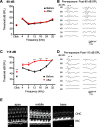
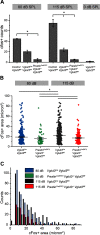
Cochlear outer hair cells (OHCs) are known to uniquely participate in auditory processing through their electromotility, and like inner hair cells, are also capable of releasing vesicular glutamate onto spiral ganglion (SG) neurons: in this case, onto the sparse Type II SG neurons. However, unlike glutamate signaling at the inner hair cell-Type I SG neuron synapse, which is robust across a wide spectrum of sound intensities, glutamate signaling at the OHC-Type II SG neuron synapse is weaker and has been hypothesized to occur only at intense, possibly damaging sound levels. Here, we tested the ability of the OHC-Type II SG pathway to signal to the brain in response to moderate, nondamaging sound (80 dB SPL) as well as to intense sound (115 dB SPL). First, we determined the VGluTs associated with OHC signaling and then confirmed the loss of glutamatergic synaptic transmission from OHCs to Type II SG neurons in KO mice using dendritic patch-clamp recordings. Next, we generated genetic mouse lines in which vesicular glutamate release occurs selectively from OHCs, and then assessed c-Fos expression in the cochlear nucleus in response to sound. From these analyses, we show, for the first time, that glutamatergic signaling at the OHC-Type II SG neuron synapse is capable of activating cochlear nucleus neurons, even at moderate sound levels.SIGNIFICANCE STATEMENT Evidence suggests that cochlear outer hair cells (OHCs) release glutamate onto Type II spiral ganglion neurons only when exposed to loud sound, and that Type II neurons are activated by tissue damage. Knowing whether moderate level sound, without tissue damage, activates this pathway has functional implications for this fundamental auditory pathway. We first determined that OHCs rely largely on VGluT3 for synaptic glutamate release. We then used a genetically modified mouse line in which OHCs, but not inner hair cells, release vesicular glutamate to demonstrate that moderate sound exposure activates cochlear nucleus neurons via the OHC-Type II spiral ganglion pathway. Together, these data indicate that glutamate signaling at the OHC-Type II afferent synapse participates in auditory function at moderate sound levels.
Keywords: Type II spiral ganglion neurons; cochlear nucleus; glutamate signaling; granule cells; outer hair cells.
Copyright © 2021 the authors.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
