Baroreceptor Modulation of the Cardiovascular System, Pain, Consciousness, and Cognition
- PMID: 33577130
- PMCID: PMC8480547
- DOI: 10.1002/cphy.c190038
Baroreceptor Modulation of the Cardiovascular System, Pain, Consciousness, and Cognition
Abstract
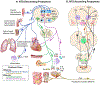
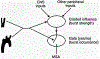
Baroreceptors are mechanosensitive elements of the peripheral nervous system that maintain cardiovascular homeostasis by coordinating the responses to external and internal environmental stressors. While it is well known that carotid and cardiopulmonary baroreceptors modulate sympathetic vasomotor and parasympathetic cardiac neural autonomic drive, to avoid excessive fluctuations in vascular tone and maintain intravascular volume, there is increasing recognition that baroreceptors also modulate a wide range of non-cardiovascular physiological responses via projections from the nucleus of the solitary tract to regions of the central nervous system, including the spinal cord. These projections regulate pain perception, sleep, consciousness, and cognition. In this article, we summarize the physiology of baroreceptor pathways and responses to baroreceptor activation with an emphasis on the mechanisms influencing cardiovascular function, pain perception, consciousness, and cognition. Understanding baroreceptor-mediated effects on cardiac and extra-cardiac autonomic activities will further our understanding of the pathophysiology of multiple common clinical conditions, such as chronic pain, disorders of consciousness (e.g., abnormalities in sleep-wake), and cognitive impairment, which may result in the identification and implementation of novel treatment modalities. © 2021 American Physiological Society. Compr Physiol 11:1373-1423, 2021.
Copyright © 2021 American Physiological Society. All rights reserved.
Figures
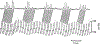
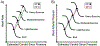
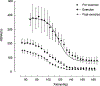



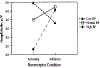


References
-
- Aars H. Effects of noradrenaline on activity in single aortic baroreceptor fibres. Acta Physiol (Oxf) 83: 335–343, 1971. - PubMed
-
- Abboud FM, and Thames MD. Interaction of cardiovascular reflexes in circulatory control. In: Handbook of Physiology - The Cardiovascular System, edited by Shepherd JT, and Abboud FM. Bethesda, MD: American Physiological Society, 1984, p. 675–753.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

