Porous Hybrids Structure between Silver Nanoparticle and Layered Double Hydroxide for Surface-Enhanced Raman Spectroscopy
- PMID: 33578775
- PMCID: PMC7916476
- DOI: 10.3390/nano11020447
Porous Hybrids Structure between Silver Nanoparticle and Layered Double Hydroxide for Surface-Enhanced Raman Spectroscopy
Abstract
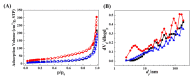
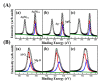
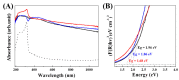
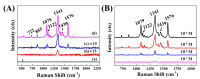
Silver nanoparticle (AgNP), in terms of antibacterial, catalytic, electronic, and optical applications, is an attractive material. Especially, when prepared to furnish sharp edge and systematic particle orientation on the substrate, AgNPs can take advantage of surface-enhanced Raman spectroscopy (SERS). In this research, we suggested a synthetic method to immobilize the AgNP on metal oxide by utilizing Ag-thiolate and layered double hydroxide (LDH) as precursor and template, respectively. The layer-by-layer structure of LDH and Ag-thiolate transformed through reductive calcination to metal oxide and AgNP array. Physicochemical characterization, including powder X-ray diffraction, N2 adsorption-desorption, microscopies, and X-ray photoelectron spectroscopy, revealed that the AgNP with sufficient crystallinity and particle gap was obtained at relatively high calcination temperature, ~600 °C. UV-vis diffusion reflectance spectroscopy showed that the calcination temperature affected particle size and electronic structure of AgNP. The prepared materials were subjected to SERS tests toward 4-nitrothiophenol (4-NTP). The sample obtained at 600 °C exhibited 50 times higher substrate enhancement factor (SEF) than the one obtained at 400 °C, suggesting that the calcination temperature was a determining parameter to enhance SERS activity in current synthetic condition.
Keywords: layered double hydroxide; porous structure; silver nanoparticle; surface-enhanced Raman spectroscopy.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
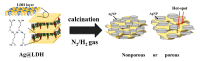



References
-
- Istiqola A., Syafiuddin A. A review of silver nanoparticles in food packaging technologies: Regulation, methods, properties, migration, and future challenges. J. Chin. Chem. Soc. 2020;67:1942–1956. doi: 10.1002/jccs.202000179. - DOI
-
- Wu Y., Yang Y., Zhang Z., Wang Z., Zhao Y., Sun L. Fabrication of cotton fabrics with durable antibacterial activities finishing by Ag nanoparticles. Text. Res. J. 2019;89:867–880. doi: 10.1177/0040517518758002. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

