Factors Associated with Hepatitis B Surface Antigen Kinetics and Responses in Pegylated Interferon Alpha-2a Monotherapy for Patients with Chronic Hepatitis B
- PMID: 33583931
- PMCID: PMC7946504
- DOI: 10.2169/internalmedicine.5432-20
Factors Associated with Hepatitis B Surface Antigen Kinetics and Responses in Pegylated Interferon Alpha-2a Monotherapy for Patients with Chronic Hepatitis B
Abstract
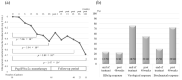
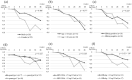
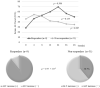
Objective Pegylated-interferon monotherapy is the standard treatment for patients with chronic hepatitis B; however, the factors associated with its therapeutic effects remain unclear. Methods Patients with chronic hepatitis B were treated with pegylated interferon α-2a for 48 weeks. We evaluated the kinetics of hepatitis B surface antigen (HBsAg) during treatment and follow-up periods and the factors associated with an HBsAg response (defined as a change in HBsAg of ≥-1 log IU/mL from baseline). Results The study population comprised 50 patients. The median baseline levels of hepatitis B virus DNA and HBsAg were 5.00 and 3.40 log IU/mL. The median values of HBsAg reduction from baseline were -0.44 (n=48), -0.41 (n=40), and -0.68 (n=11) log IU/mL at the end of treatment and at 48 and 144 weeks post-treatment, respectively. The rates of HBsAg response were 24.0% and 22.5% at the end of treatment and at 48 weeks post-treatment, respectively. A multivariate analysis identified HBsAg <3.00 log IU/mL as an independent baseline factor contributing to the HBsAg response at the end of treatment and 48 weeks post-treatment (p=1.07×10-2 and 4.42×10-2, respectively). There were significant differences in the reduction of the HBsAg levels at 12 weeks of treatment and in the incidence of serum ALT increase during treatment between patients with and without an HBsAg response. Conclusion These findings suggest that the baseline HBsAg level, HBsAg kinetics at 12 weeks of treatment, and ALT increase during treatment are important factors contributing to the HBsAg response in pegylated interferon α-2a monotherapy for patients with chronic hepatitis B.
Keywords: HBsAg response; hepatitis B surface antigen (HBsAg) kinetics; pegylated interferon alpha-2a monotherapy.
Conflict of interest statement
Figures

Similar articles
-
Kinetics of Hepatitis B Surface Antigen Level in Chronic Hepatitis B Patients who Achieved Hepatitis B Surface Antigen Loss during Pegylated Interferon Alpha-2a Treatment.Chin Med J (Engl). 2017 Mar 5;130(5):559-565. doi: 10.4103/0366-6999.200554. Chin Med J (Engl). 2017. PMID: 28229987 Free PMC article.
-
[HBsAg loss with Pegylated-interferon alfa-2a in hepatitis B patients with partial response to nucleos(t)-ide analog: new switch study].Zhonghua Gan Zang Bing Za Zhi. 2018 Oct 20;26(10):756-764. doi: 10.3760/cma.j.issn.1007-3418.2018.10.005. Zhonghua Gan Zang Bing Za Zhi. 2018. PMID: 30481882 Clinical Trial. Chinese.
-
Effect of switching from treatment with nucleos(t)ide analogs to pegylated interferon α-2a on virological and serological responses in chronic hepatitis B patients.World J Gastroenterol. 2016 Dec 14;22(46):10210-10218. doi: 10.3748/wjg.v22.i46.10210. World J Gastroenterol. 2016. PMID: 28028369 Free PMC article. Clinical Trial.
-
Hepatitis D Virus and HBsAg Dynamics in the era of new Antiviral Treatments.Curr Gastroenterol Rep. 2023 Dec;25(12):401-412. doi: 10.1007/s11894-023-00901-9. Epub 2023 Oct 11. Curr Gastroenterol Rep. 2023. PMID: 37819559 Free PMC article. Review.
-
[Progress in the clinical cure of the population of inactive hepatitis B surface antigen carriers].Zhonghua Gan Zang Bing Za Zhi. 2023 Dec 20;31(12):1336-1339. doi: 10.3760/cma.j.cn501113-20231116-00201. Zhonghua Gan Zang Bing Za Zhi. 2023. PMID: 38253081 Review. Chinese.
Cited by
-
Cases of Rapid Hepatitis B Surface Antigen Reduction after COVID-19 Vaccination.Intern Med. 2023 Jan 1;62(1):51-57. doi: 10.2169/internalmedicine.0842-22. Epub 2022 Oct 19. Intern Med. 2023. PMID: 36261382 Free PMC article.
-
Predicting clinical outcomes in chronic hepatitis B patients receiving nucleoside analogues and pegylated interferon alpha: a hematochemical and clinical analysis.BMC Infect Dis. 2024 Oct 13;24(1):1149. doi: 10.1186/s12879-024-10057-0. BMC Infect Dis. 2024. PMID: 39396949 Free PMC article.
References
-
- World Health Organization. Global Hepatitis Report 2017 [Internet]. [cited 2019 Feb 8]. Available from: https://apps.who.int/iris/bitstream/handle/10665/255016/9789241565455-en...
-
- Dienstag JL. Hepatitis B virus infection. N Engl J Med 359: 1486-1500, 2008. - PubMed
-
- Chan HL, Sung JJ. Hepatocellular carcinoma and hepatitis B virus. Semin Liver Dis 26: 153-161, 2006. - PubMed
-
- Simonetti J, Bulkow L, McMahon BJ, et al. . Clearance of hepatitis B surface antigen and risk of hepatocellular carcinoma in a cohort chronically infected with hepatitis B virus. Hepatology 51: 1531-1537, 2010. - PubMed
-
- Tseng TC, Liu CJ, Yang HC, et al. . High levels of hepatitis B surface antigen increase risk of hepatocellular carcinoma in patients with low HBV load. Gastroenterology 142: 1140-1149. e3; quiz e13-e4, 2012. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

