Rv1717 Is a Cell Wall - Associated β-Galactosidase of Mycobacterium tuberculosis That Is Involved in Biofilm Dispersion
- PMID: 33584576
- PMCID: PMC7873859
- DOI: 10.3389/fmicb.2020.611122
Rv1717 Is a Cell Wall - Associated β-Galactosidase of Mycobacterium tuberculosis That Is Involved in Biofilm Dispersion
Abstract
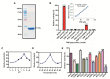
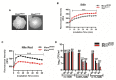
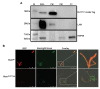
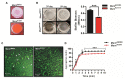
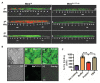
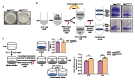
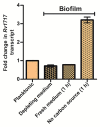
Understanding the function of conserved hypothetical protein (CHP)s expressed by a pathogen in the infected host can lead to better understanding of its pathogenesis. The present work describes the functional characterization of a CHP, Rv1717 of Mycobacterium tuberculosis (Mtb). Rv1717 has been previously reported to be upregulated in TB patient lungs. Rv1717 belongs to the cupin superfamily of functionally diverse proteins, several of them being carbohydrate handling proteins. Bioinformatic analysis of the amino acid sequence revealed similarity to glycosyl hydrolases. Enzymatic studies with recombinant Rv1717 purified from Escherichia coli showed that the protein is a β-D-galactosidase specific for pyranose form rather than the furanose form. We expressed the protein in Mycobacterium smegmatis (Msm), which lacks its ortholog. In Msm Rv1717 , the protein was found to localize to the cell wall (CW) with a preference to the poles. Msm Rv1717 showed significant changes in colony morphology and cell surface properties. Most striking observation was its unusual Congo red colony morphotype, reduced ability to form biofilms, pellicles and autoagglutinate. Exogenous Rv1717 not only prevented biofilm formation in Msm, but also degraded preformed biofilms, suggesting that its substrate likely exists in the exopolysaccharides of the biofilm matrix. Presence of galactose in the extracellular polymeric substance (EPS) has not been reported before and hence we used the galactose-specific Wisteria floribunda lectin (WFL) to test the same. The lectin extensively bound to Msm and Mtb EPS, but not the bacterium per se. Purified Rv1717 also hydrolyzed exopolysaccharides extracted from Msm biofilm. Eventually, to decipher its role in Mtb, we downregulated its expression and demonstrate that the strain is unable to disperse from in vitro biofilms, unlike the wild type. Biofilms exposed to carbon starvation showed a sudden upregulation of Rv1717 transcripts supporting the potential role of Rv1717 in Mtb dispersing from a deteriorating biofilm.
Keywords: biofilm; biofilm dispersion; extracellular polymeric substance; galactosidase; mycobacteria.
Copyright © 2021 Bharti, Maurya, Venugopal, Singh, Akhtar and Krishnan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources

