A critical residue in the α1M2-M3 linker regulating mammalian GABAA receptor pore gating by diazepam
- PMID: 33591271
- PMCID: PMC7899671
- DOI: 10.7554/eLife.64400
A critical residue in the α1M2-M3 linker regulating mammalian GABAA receptor pore gating by diazepam
Abstract
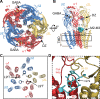
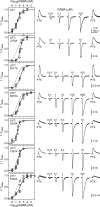
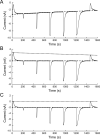
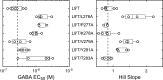
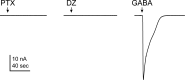
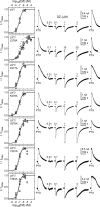
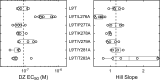
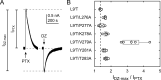
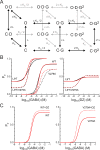
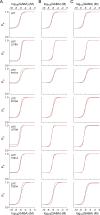
Benzodiazepines (BZDs) are a class of widely prescribed psychotropic drugs that modulate activity of GABAA receptors (GABAARs), neurotransmitter-gated ion channels critical for synaptic transmission. However, the physical basis of this modulation is poorly understood. We explore the role of an important gating domain, the α1M2-M3 linker, in linkage between the BZD site and pore gate. To probe energetics of this coupling without complication from bound agonist, we use a gain of function mutant (α1L9'Tβ2γ2L) directly activated by BZDs. We identify a specific residue whose mutation (α1V279A) more than doubles the energetic contribution of the BZD positive modulator diazepam (DZ) to pore opening and also enhances DZ potentiation of GABA-evoked currents in a wild-type background. In contrast, other linker mutations have little effect on DZ efficiency, but generally impair unliganded pore opening. Our observations reveal an important residue regulating BZD-pore linkage, thereby shedding new light on the molecular mechanism of these drugs.
Keywords: GABAA receptor; M2-M3 linker; allosteric modulator; benzodiazepine; diazepam; molecular biophysics; structural biology; xenopus.
© 2021, Nors et al.
Conflict of interest statement
JN, SG, MG No competing interests declared
Figures





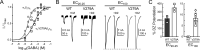
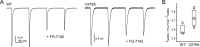
Similar articles
-
Benzodiazepine Modulation of GABAA Receptors: A Mechanistic Perspective.Biomolecules. 2022 Nov 30;12(12):1784. doi: 10.3390/biom12121784. Biomolecules. 2022. PMID: 36551212 Free PMC article. Review.
-
GABAA receptor subunit M2-M3 linkers have asymmetric roles in pore gating and diazepam modulation.Biophys J. 2024 Jul 16;123(14):2085-2096. doi: 10.1016/j.bpj.2024.02.016. Epub 2024 Feb 22. Biophys J. 2024. PMID: 38400541 Free PMC article.
-
Structural mechanisms underlying benzodiazepine modulation of the GABA(A) receptor.J Neurosci. 2008 Mar 26;28(13):3490-9. doi: 10.1523/JNEUROSCI.5727-07.2008. J Neurosci. 2008. PMID: 18367615 Free PMC article.
-
Positive modulation of synaptic and extrasynaptic GABAA receptors by an antagonist of the high affinity benzodiazepine binding site.Neuropharmacology. 2015 Aug;95:459-67. doi: 10.1016/j.neuropharm.2015.04.027. Epub 2015 May 9. Neuropharmacology. 2015. PMID: 25963418
-
Identification of transduction elements for benzodiazepine modulation of the GABA(A) receptor: three residues are required for allosteric coupling.J Neurosci. 1999 Dec 1;19(23):10213-20. doi: 10.1523/JNEUROSCI.19-23-10213.1999. J Neurosci. 1999. PMID: 10575018 Free PMC article.
Cited by
-
State-dependent energetics of GABAA receptor modulators.Biophys J. 2024 Jul 16;123(14):1903-1906. doi: 10.1016/j.bpj.2024.01.035. Epub 2024 Feb 1. Biophys J. 2024. PMID: 38303510 Free PMC article. No abstract available.
-
Shisa7-Dependent Regulation of GABAA Receptor Single-Channel Gating Kinetics.J Neurosci. 2022 Nov 23;42(47):8758-8766. doi: 10.1523/JNEUROSCI.0510-22.2022. Epub 2022 Oct 10. J Neurosci. 2022. PMID: 36216503 Free PMC article.
-
A single main-chain hydrogen bond required to keep GABAA receptors closed.Nat Commun. 2025 Jul 3;16(1):6107. doi: 10.1038/s41467-025-61447-0. Nat Commun. 2025. PMID: 40603861 Free PMC article.
-
Benzodiazepine Modulation of GABAA Receptors: A Mechanistic Perspective.Biomolecules. 2022 Nov 30;12(12):1784. doi: 10.3390/biom12121784. Biomolecules. 2022. PMID: 36551212 Free PMC article. Review.
-
Allosteric modulation of α1β3γ2 GABAA receptors by farnesol through the neurosteroid sites.Biophys J. 2023 Mar 7;122(5):849-867. doi: 10.1016/j.bpj.2023.01.032. Epub 2023 Jan 31. Biophys J. 2023. PMID: 36721367 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

