BK Channel Regulation of Afterpotentials and Burst Firing in Cerebellar Purkinje Neurons
- PMID: 33593855
- PMCID: PMC8018884
- DOI: 10.1523/JNEUROSCI.0192-20.2021
BK Channel Regulation of Afterpotentials and Burst Firing in Cerebellar Purkinje Neurons
Abstract
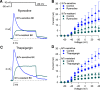
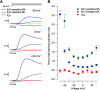
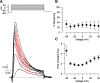
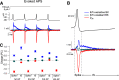
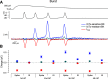
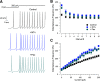
BK calcium-activated potassium channels have complex kinetics because they are activated by both voltage and cytoplasmic calcium. The timing of BK activation and deactivation during action potentials determines their functional role in regulating firing patterns but is difficult to predict a priori. We used action potential clamp to characterize the kinetics of voltage-dependent calcium current and BK current during action potentials in Purkinje neurons from mice of both sexes, using acutely dissociated neurons that enabled rapid voltage clamp at 37°C. With both depolarizing voltage steps and action potential waveforms, BK current was entirely dependent on calcium entry through voltage-dependent calcium channels. With voltage steps, BK current greatly outweighed the triggering calcium current, with only a brief, small net inward calcium current before Ca-activated BK current dominated the total Ca-dependent current. During action potential waveforms, although BK current activated with only a short (∼100 μs) delay after calcium current, the two currents were largely separated, with calcium current flowing during the falling phase of the action potential and most BK current flowing over several milliseconds after repolarization. Step depolarizations activated both an iberiotoxin-sensitive BK component with rapid activation and deactivation kinetics and a slower-gating iberiotoxin-resistant component. During action potential firing, however, almost all BK current came from the faster-gating iberiotoxin-sensitive channels, even during bursts of action potentials. Inhibiting BK current had little effect on action potential width or a fast afterhyperpolarization but converted a medium afterhyperpolarization to an afterdepolarization and could convert tonic firing of single action potentials to burst firing.SIGNIFICANCE STATEMENT BK calcium-activated potassium channels are widely expressed in central neurons. Altered function of BK channels is associated with epilepsy and other neuronal disorders, including cerebellar ataxia. The functional role of BK in regulating neuronal firing patterns is highly dependent on the context of other channels and varies widely among different types of neurons. Most commonly, BK channels are activated during action potentials and help produce a fast afterhyperpolarization. We find that in Purkinje neurons BK current flows primarily after the fast afterhyperpolarization and helps to prevent a later afterdepolarization from producing rapid burst firing, enabling typical regular tonic firing.
Keywords: KCa1.1; action potential clamp; calcium channel; cerebellum; iberiotoxin; paxilline.
Copyright © 2021 the authors.
Figures







Similar articles
-
Iberiotoxin-sensitive large conductance Ca2+ -dependent K+ (BK) channels regulate the spike configuration in the burst firing of cerebellar Purkinje neurons.Brain Res. 2008 May 30;1212:1-8. doi: 10.1016/j.brainres.2008.03.030. Epub 2008 Mar 27. Brain Res. 2008. PMID: 18439989
-
Differential Regulation of Action Potential Shape and Burst-Frequency Firing by BK and Kv2 Channels in Substantia Nigra Dopaminergic Neurons.J Neurosci. 2015 Dec 16;35(50):16404-17. doi: 10.1523/JNEUROSCI.5291-14.2015. J Neurosci. 2015. PMID: 26674866 Free PMC article.
-
Iberiotoxin-sensitive and -insensitive BK currents in Purkinje neuron somata.J Neurophysiol. 2013 May;109(10):2528-41. doi: 10.1152/jn.00127.2012. Epub 2013 Feb 27. J Neurophysiol. 2013. PMID: 23446695 Free PMC article.
-
Current understanding of iberiotoxin-resistant BK channels in the nervous system.Front Physiol. 2014 Oct 9;5:382. doi: 10.3389/fphys.2014.00382. eCollection 2014. Front Physiol. 2014. PMID: 25346692 Free PMC article. Review.
-
Calcium-activated potassium channels in adrenal chromaffin cells.Ion Channels. 1996;4:261-301. doi: 10.1007/978-1-4899-1775-1_7. Ion Channels. 1996. PMID: 8744211 Review.
Cited by
-
Evidence for Dual Activation of IK(M) and IK(Ca) Caused by QO-58 (5-(2,6-Dichloro-5-fluoropyridin-3-yl)-3-phenyl-2-(trifluoromethyl)-1H-pyrazolol[1,5-a]pyrimidin-7-one).Int J Mol Sci. 2022 Jun 24;23(13):7042. doi: 10.3390/ijms23137042. Int J Mol Sci. 2022. PMID: 35806047 Free PMC article.
-
Knockdown of the Non-canonical Wnt Gene Prickle2 Leads to Cerebellar Purkinje Cell Abnormalities While Cerebellar-Mediated Behaviors Remain Intact.Cerebellum. 2024 Oct;23(5):1741-1753. doi: 10.1007/s12311-023-01648-9. Epub 2024 Jan 2. Cerebellum. 2024. PMID: 38165577 Free PMC article.
-
α2δ-2 is required for depolarization-induced suppression of excitation in Purkinje cells.J Physiol. 2022 Jan;600(1):111-122. doi: 10.1113/JP282438. Epub 2021 Dec 3. J Physiol. 2022. PMID: 34783012 Free PMC article.
-
Estradiol elicits distinct firing patterns in arcuate nucleus kisspeptin neurons of females through altering ion channel conductances.bioRxiv [Preprint]. 2024 Sep 3:2024.02.20.581121. doi: 10.1101/2024.02.20.581121. bioRxiv. 2024. Update in: Elife. 2024 Dec 13;13:RP96691. doi: 10.7554/eLife.96691. PMID: 38915596 Free PMC article. Updated. Preprint.
-
[Expression changes of NaV channel subunits correlate with developmental maturation of electrophysiological characteristics of rat cerebellar Purkinje neurons].Nan Fang Yi Ke Da Xue Xue Bao. 2023 Jul 20;43(7):1102-1109. doi: 10.12122/j.issn.1673-4254.2023.07.06. Nan Fang Yi Ke Da Xue Xue Bao. 2023. PMID: 37488792 Free PMC article. Chinese.
References
-
- Ait Ouares K, Filipis L, Tzilivaki A, Poirazi P, Canepari M (2019) Two distinct sets of Ca2+ and K+ channels are activated at different membrane potentials by the climbing fiber synaptic potential in Purkinje neuron dendrites. J Neurosci 39:1969–1981. 10.1523/JNEUROSCI.2155-18.2018 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
