Brachypodium Phenylalanine Ammonia Lyase (PAL) Promotes Antiviral Defenses against Panicum mosaic virus and Its Satellites
- PMID: 33593968
- PMCID: PMC8545123
- DOI: 10.1128/mBio.03518-20
Brachypodium Phenylalanine Ammonia Lyase (PAL) Promotes Antiviral Defenses against Panicum mosaic virus and Its Satellites
Abstract
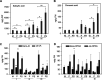
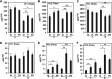
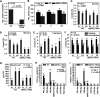
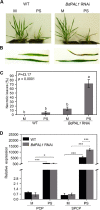
Brachypodium distachyon has recently emerged as a premier model plant for monocot biology, akin to Arabidopsis thaliana We previously reported genome-wide transcriptomic and alternative splicing changes occurring in Brachypodium during compatible infections with Panicum mosaic virus (PMV) and its satellite virus (SPMV). Here, we dissected the role of Brachypodium phenylalanine ammonia lyase 1 (PAL1), a key enzyme for phenylpropanoid and salicylic acid (SA) biosynthesis and the induction of plant defenses. Targeted metabolomics profiling of PMV-infected and PMV- plus SPMV-infected (PMV/SPMV) Brachypodium plants revealed enhanced levels of multiple defense-related hormones and metabolites such as cinnamic acid, SA, and fatty acids and lignin precursors during disease progression. The virus-induced accumulation of SA and lignin was significantly suppressed upon knockdown of B. distachyonPAL1 (BdPAL1) using RNA interference (RNAi). The compromised SA accumulation in PMV/SPMV-infected BdPAL1 RNAi plants correlated with weaker induction of multiple SA-related defense gene markers (pathogenesis related 1 [PR-1], PR-3, PR-5, and WRKY75) and enhanced susceptibility to PMV/SPMV compared to that of wild-type (WT) plants. Furthermore, exogenous application of SA alleviated the PMV/SPMV necrotic disease phenotypes and delayed plant death caused by single and mixed infections. Together, our results support an antiviral role for BdPAL1 during compatible host-virus interaction, perhaps as a last resort attempt to rescue the infected plant.IMPORTANCE Although the role of plant defense mechanisms against viruses are relatively well studied in dicots and in incompatible plant-microbe interactions, studies of their roles in compatible interactions and in grasses are lagging behind. In this study, we leveraged the emerging grass model Brachypodium and genetic resources to dissect Panicum mosaic virus (PMV)- and its satellite virus (SPMV)-compatible grass-virus interactions. We found a significant role for PAL1 in the production of salicylic acid (SA) in response to PMV/SPMV infections and that SA is an essential component of the defense response preventing the plant from succumbing to viral infection. Our results suggest a convergent role for the SA defense pathway in both compatible and incompatible plant-virus interactions and underscore the utility of Brachypodium for grass-virus biology.
Keywords: bioenergy; defense hormones; grasses; metabolic pathways; plant-virus interactions.
Figures

References
-
- Yang H, Gou X, He K, Xi D, Du J, Lin H, Li J. 2010. BAK1 and BKK1 in Arabidopsis thaliana confer reduced susceptibility to Turnip crinkle virus. Eur J Plant Pathol 127:149–156. doi: 10.1007/s10658-010-9581-5. - DOI
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
