LACTB induced apoptosis of oxaliplatin-resistant gastric cancer through regulating autophagy-mediated mitochondrial apoptosis pathway
- PMID: 33594312
- PMCID: PMC7868839
LACTB induced apoptosis of oxaliplatin-resistant gastric cancer through regulating autophagy-mediated mitochondrial apoptosis pathway
Abstract
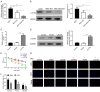
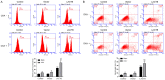
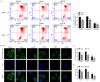
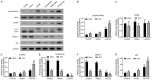
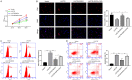
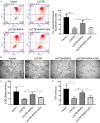
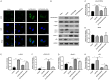
Oxaliplatin (OXA), as a third-generation platinum anticancer drug, is a treatment drug for gastric cancer (GC). However, OXA resistance has become the main reason for OXA treatment failure. Serine beta-lactamase-like protein (LACTB), acts as a mitochondrial protein, can affect multiple cancer processes. Here, we aimed to investigate the function and mechanism of LACTB in OXA-resistant GC. After LACTB overexpression or autophagy activator (RAPA) treatment, cell proliferation, reactive oxygen species (ROS), apoptosis, mitochondrial dysfunction were evaluated through CCK-8 assay, Edu staining, flow cytometry and immunofluorescence assay. Moreover, DNA double-stranded damage and autophagy-related proteins were examined via western blot. We revealed that LACTB was downregulated in OXA-resistant MGC-803 cells, and overexpression of LACTB reduced the resistance of GC cells to OXA. Besides, our results uncovered that overexpression of LACTB induced apoptosis, reduced the mitochondrial membrane potential (MMP) and accelerated ROS accumulation in OXA-resistant MGC-803 (MGC-803/OXA) cells. Meanwhile, we verified that overexpression of LACTB decreased glucose uptake and ATP synthesis, induced mitochondria and DNA damages, and inhibited autophagy of MGC-803/OXA cells. Furthermore, our results certified that RAPA could weaken the function of LACTB on apoptosis and mitochondrial morphology and function in OXA-resistant MGC-803 cells with OXA treatment. Therefore, we demonstrated that LACTB could attenuate the resistance of MGC-803/OXA cells to OXA through autophagy-mediated mitochondrial morphological changes, mitochondrial dysfunction, and apoptosis, suggesting that LACTB, functions as a suppressor, is conducive to the therapy of OXA-resistant GC.
Keywords: Gastric cancer; apoptosis; autophagy; mitochondrial dysfunction; oxaliplatin; serine beta-lactamase-like protein.
AJTR Copyright © 2021.
Conflict of interest statement
None.
Figures
Similar articles
-
Unveiling the Function of the Mitochondrial Filament-Forming Protein LACTB in Lipid Metabolism and Cancer.Cells. 2022 May 20;11(10):1703. doi: 10.3390/cells11101703. Cells. 2022. PMID: 35626737 Free PMC article. Review.
-
PCBP1 protects bladder cancer cells from mitochondria injury and ferroptosis by inducing LACTB mRNA degradation.Mol Carcinog. 2023 Jul;62(7):907-919. doi: 10.1002/mc.23533. Epub 2023 May 8. Mol Carcinog. 2023. PMID: 37157950
-
Mitochondrial ROS accumulation inhibiting JAK2/STAT3 pathway is a critical modulator of CYT997-induced autophagy and apoptosis in gastric cancer.J Exp Clin Cancer Res. 2020 Jun 23;39(1):119. doi: 10.1186/s13046-020-01621-y. J Exp Clin Cancer Res. 2020. PMID: 32576206 Free PMC article.
-
The long non-coding RNA DDX11-AS1 facilitates cell progression and oxaliplatin resistance via regulating miR-326/IRS1 axis in gastric cancer.Eur Rev Med Pharmacol Sci. 2020 Mar;24(6):3049-3061. doi: 10.26355/eurrev_202003_20669. Eur Rev Med Pharmacol Sci. 2020. PMID: 32271422
-
The Pivotal Role of LACTB in the Process of Cancer Development.Int J Mol Sci. 2025 Feb 1;26(3):1279. doi: 10.3390/ijms26031279. Int J Mol Sci. 2025. PMID: 39941048 Free PMC article. Review.
Cited by
-
Knockdown of SLC7A5 inhibits malignant progression and attenuates oxaliplatin resistance in gastric cancer by suppressing glycolysis.Mol Med. 2025 Mar 25;31(1):115. doi: 10.1186/s10020-025-01175-9. Mol Med. 2025. PMID: 40133832 Free PMC article.
-
Docosahexaenoic Acid Counteracts the Hypoxic-Induced Inflammatory and Metabolic Alterations in 3T3-L1 Adipocytes.Nutrients. 2022 Nov 1;14(21):4600. doi: 10.3390/nu14214600. Nutrients. 2022. PMID: 36364860 Free PMC article.
-
CRISPR-based knockout screening identifies the loss of MIEF2 to enhance oxaliplatin resistance in colorectal cancer through inhibiting the mitochondrial apoptosis pathway.Front Oncol. 2022 Aug 29;12:881487. doi: 10.3389/fonc.2022.881487. eCollection 2022. Front Oncol. 2022. PMID: 36106106 Free PMC article.
-
LACTB suppresses carcinogenesis in lung cancer and regulates the EMT pathway.Exp Ther Med. 2022 Mar;23(3):247. doi: 10.3892/etm.2022.11172. Epub 2022 Jan 28. Exp Ther Med. 2022. PMID: 35222724 Free PMC article.
-
Unveiling the Function of the Mitochondrial Filament-Forming Protein LACTB in Lipid Metabolism and Cancer.Cells. 2022 May 20;11(10):1703. doi: 10.3390/cells11101703. Cells. 2022. PMID: 35626737 Free PMC article. Review.
References
-
- Fritsch R, Hoeppner J. Oxaliplatin in perioperative chemotherapy for gastric and gastroesophageal junction (GEJ) adenocarcinoma. Expert Rev Gastroenterol Hepatol. 2019;13:285–291. - PubMed
-
- Osawa H, Yamada K, Kuroda Y. Trastuzumab combined oxaliplatin and S-1 therapy demonstrated pathological complete response of synchronous liver metastasis of gastric cancer. Cell Mol Med. 2017;3:1.
-
- Petrioli R, Francini E, Cherri S, Marrelli D, Rovello F, Fiaschi AI, Miano ST, Savelli V, Calomino N, Farsi M, Vernillo R, Francini G. Feasibility of modified docetaxel, oxaliplatin, capecitabine followed by capecitabine as maintenance chemotherapy as first-line therapy for patients with metastatic gastric or gastroesophageal cancer. Anticancer Drugs. 2020;31:292–297. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
