Clarithromycin impairs tissue-resident memory and Th17 responses to macrolide-resistant Streptococcus pneumoniae infections
- PMID: 33595670
- PMCID: PMC8164591
- DOI: 10.1007/s00109-021-02039-5
Clarithromycin impairs tissue-resident memory and Th17 responses to macrolide-resistant Streptococcus pneumoniae infections
Abstract
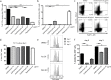
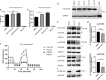
The increasing prevalence of antimicrobial resistance in pathogens is a growing public health concern, with the potential to compromise the success of infectious disease treatments in the future. Particularly, the number of infections by macrolide antibiotics-resistant Streptococcus pneumoniae is increasing. We show here that Clarithromycin impairs both the frequencies and number of interleukin (IL)-17 producing T helper (Th) 17 cells within the lungs of mice infected with a macrolide-resistant S. pneumoniae serotype 15A strain. Subsequently, the tissue-resident memory CD4+ T cell (Trm) response to a consecutive S. pneumoniae infection was impaired. The number of lung resident IL-17+ CD69+ Trm was diminished upon Clarithromycin treatment during reinfection. Mechanistically, Clarithromycin attenuated phosphorylation of the p90-S6-kinase as part of the ERK pathway in Th17 cells. Moreover, a strong increase in the mitochondrial-mediated maximal respiratory capacity was observed, while mitochondrial protein translation and mTOR sisgnaling were unimpaired. Therefore, treatment with macrolide antibiotics may favor the spread of antimicrobial-resistant pathogens not only by applying a selection pressure but also by decreasing the natural T cell immune response. Clinical administration of macrolide antibiotics as standard therapy procedure during initial hospitalization should be reconsidered accordingly and possibly be withheld until microbial resistance is determined. KEY MESSAGES: • Macrolide-resistant S. pneumoniae infection undergoes immunomodulation by Clarithromycin • Clarithromycin treatment hinders Th17 and tissue-resident memory responses • Macrolide antibiotics impair Th17 differentiation in vitro by ERK-pathway inhibition.
Keywords: Anti-microbial resistance; Clarithromycin; Macrolide antibiotics; Streptococcus pneumoniae; Th17 cells; Tissue-resident memory T cells.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



Similar articles
-
Macrolide and ketolide resistance with Streptococcus pneumoniae.Med Clin North Am. 2006 Nov;90(6):1109-24. doi: 10.1016/j.mcna.2006.07.010. Med Clin North Am. 2006. PMID: 17116439 Review.
-
Phenotypic and molecular characterization of macrolide resistance mechanisms among Streptococcus pneumoniae isolated in Tunisia.J Med Microbiol. 2020 Apr;69(4):505-520. doi: 10.1099/jmm.0.001151. J Med Microbiol. 2020. PMID: 32159507
-
Macrolide Resistance in Streptococcus pneumoniae.Front Cell Infect Microbiol. 2016 Sep 21;6:98. doi: 10.3389/fcimb.2016.00098. eCollection 2016. Front Cell Infect Microbiol. 2016. PMID: 27709102 Free PMC article. Review.
-
Streptococcus pneumoniae bacteraemia: pharmacodynamic correlations with outcome and macrolide resistance--a controlled study.Int J Antimicrob Agents. 2007 Sep;30(3):264-9. doi: 10.1016/j.ijantimicag.2007.04.013. Epub 2007 Jun 22. Int J Antimicrob Agents. 2007. PMID: 17587549
-
Antimicrobial and immunomodulatory effect of clarithromycin on macrolide-resistant Mycoplasma pneumoniae.J Med Microbiol. 2010 Jun;59(Pt 6):693-701. doi: 10.1099/jmm.0.014191-0. Epub 2010 Mar 11. J Med Microbiol. 2010. PMID: 20223900
Cited by
-
Tissue-resident immune cells: from defining characteristics to roles in diseases.Signal Transduct Target Ther. 2025 Jan 17;10(1):12. doi: 10.1038/s41392-024-02050-5. Signal Transduct Target Ther. 2025. PMID: 39820040 Free PMC article. Review.
-
Characterizing the influence of various antimicrobials used for metaphylaxis against bovine respiratory disease on host transcriptome responses.Front Vet Sci. 2023 Oct 5;10:1272940. doi: 10.3389/fvets.2023.1272940. eCollection 2023. Front Vet Sci. 2023. PMID: 37869487 Free PMC article.
-
Pneumolysin as a target for new therapies against pneumococcal infections: A systematic review.PLoS One. 2023 Mar 22;18(3):e0282970. doi: 10.1371/journal.pone.0282970. eCollection 2023. PLoS One. 2023. PMID: 36947540 Free PMC article.
-
Tissue-resident immunity in the lung: a first-line defense at the environmental interface.Semin Immunopathol. 2022 Nov;44(6):827-854. doi: 10.1007/s00281-022-00964-2. Epub 2022 Oct 28. Semin Immunopathol. 2022. PMID: 36305904 Free PMC article. Review.
References
-
- O’Brien KL, Wolfson LJ, Watt JP, et al. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet. 2009;374:893–902. - PubMed
-
- Troeger CE, Khalil IA, Blacker BF, Biehl MH, Albertson SB, Zimsen SRM, Rao PC, Abate D, Admasie A, Ahmadi A, Ahmed MLCB, Akal CG, Alahdab F, Alam N, Alene KA, Alipour V, Aljunid SM, al-Raddadi RM, Alvis-Guzman N, Amini S, Anjomshoa M, Antonio CAT, Arabloo J, Aremu O, Atalay HT, Atique S, Avokpaho EFGA, Awad S, Awasthi A, Badawi A, Balakrishnan K, Banoub JAM, Barac A, Bassat Q, Bedi N, Bennett DA, Bhattacharyya K, Bhutta ZA, Bijani A, Bills CB, Car J, Carvalho F, Castañeda-Orjuela CA, Causey K, Christopher DJ, Cohen AJ, Dandona L, Dandona R, Daryani A, Demeke FM, Djalalinia S, Dubey M, Dubljanin E, Duken EE, el Sayed Zaki M, Endries AY, Fernandes E, Fischer F, Frostad J, Fullman N, Gardner WM, Geta B, Ghadiri K, Gorini G, Goulart AC, Guo Y, Hailu GB, Haj-Mirzaian A, Haj-Mirzaian A, Hamidi S, Hassen HY, Hoang CL, Horita N, Hostiuc M, Hussain Z, Irvani SSN, James SL, Jha RP, Jonas JB, Karch A, Kasaeian A, Kassa TD, Kassebaum NJ, Kefale AT, Khader YS, Khan EA, Khan G, Khan MN, Khang YH, Khoja AT, Kimokoti RW, Kisa A, Kisa S, Kissoon N, Knibbs LD, Kochhar S, Kosen S, Koul PA, Koyanagi A, Kuate Defo B, Kumar GA, Lal DK, Leshargie CT, Lewycka S, Li S, Lodha R, Macarayan ERK, Majdan M, Mamun AA, Manguerra H, Mehta V, Melese A, Memish ZA, Mengistu DT, Meretoja TJ, Mestrovic T, Miazgowski B, Mirrakhimov EM, Moazen B, Mohammad KA, Mohammed S, Monasta L, Moore CE, Morawska L, Mosser JF, Mousavi SM, Murthy S, Mustafa G, Nazari J, Nguyen CT, Nguyen HLT, Nguyen LH, Nguyen SH, Nielsen KR, Nisar MI, Nixon MR, Ogbo FA, Okoro A, Olagunju AT, Olagunju TO, Oren E, Ortiz JR, P A M, Pakhale S, Postma MJ, Qorbani M, Quansah R, Rafiei A, Rahim F, Rahimi-Movaghar V, Rai RK, Reitsma MB, Rezai MS, Rezapour A, Rios-Blancas MJ, Ronfani L, Rothenbacher D, Rubino S, Saleem Z, Sambala EZ, Samy AM, Santric Milicevic MM, Sarmiento-Suárez R, Sartorius B, Savic M, Sawhney M, Saxena S, Sbarra A, Seyedmousavi S, Shaikh MA, Sheikh A, Shigematsu M, Smith DL, Sreeramareddy CT, Stanaway JD, Sufiyan M'B, Temsah MH, Tessema B, Tran BX, Tran KB, Tsadik AG, Ullah I, Updike RL, Vasankari TJ, Veisani Y, Wada FW, Waheed Y, Welgan K, Wiens KE, Wiysonge CS, Yimer EM, Yonemoto N, Zaidi Z, Zar HJ, Lim SS, Vos T, Mokdad AH, Murray CJL, Kyu HH, Hay SI, Reiner RC. Quantifying risks and interventions that have affected the burden of lower respiratory infections among children younger than 5 years: an analysis for the Global Burden of Disease Study 2017. Lancet Infect Dis. 2020;20:60–79. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

