Ex vivo modelling of PD-1/PD-L1 immune checkpoint blockade under acute, chronic, and exhaustion-like conditions of T-cell stimulation
- PMID: 33597595
- PMCID: PMC7889918
- DOI: 10.1038/s41598-021-83612-3
Ex vivo modelling of PD-1/PD-L1 immune checkpoint blockade under acute, chronic, and exhaustion-like conditions of T-cell stimulation
Abstract
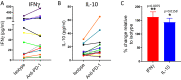
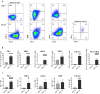
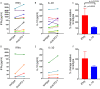
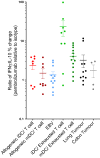
Blockade of PD-1/PD-L1 interactions is proving an exciting, durable therapeutic modality in a range of cancers whereby T cells are released from checkpoint inhibition to revive their inherent anti-tumour activity. Here we have studied various ways to model ex vivo T cell function in order to compare the impact of the clinically utilised anti-PD-1 antibody, pembrolizumab (Keytruda) on the activation of human T cells: focussing on the release of pro-inflammatory IFNγ and anti-inflammatory IL-10 to assess functionality. Firstly, we investigated the actions of pembrolizumab in an acute model of T-cell activation with either immature or mature allogeneic dendritic cells (DCs); pembrolizumab enhanced IFNγ and IL-10 release from purified CD4+ T-cells in the majority of donors with a bias towards pro-inflammatory cytokine release. Next, we modelled the impact of pembrolizumab in settings of more chronic T-cell activation. In a 7-day antigen-specific response to EBV peptides, the presence of pembrolizumab resulted in a relatively modest increase in both IFNγ and IL-10 release. Where pembrolizumab was assessed against long-term stimulated CD4+ cells that had up-regulated the exhaustion markers TIM-3 and PD-1, there was a highly effective enhancement of the otherwise exhausted response to allogeneic DCs with respect to IFNγ production. By contrast, the restoration of IL-10 production was considerably more limited. Finally, to assess a direct clinical relevance we investigated the consequence of PD-1/PD-L1 blockade in the disease setting of dissociated cells from lung and colon carcinomas responding to allogeneic DCs: here, pembrolizumab once more enhanced IFNγ production from the majority of tumour preparations whereas, again, the increase in IL-10 release was modest at best. In conclusion, we have shown that the contribution of PD-1-revealed by using a canonical blocking antibody to interrupt its interaction with PD-L1-to the production of an exemplar pro- and anti-inflammatory cytokine, respectively, depends in magnitude and ratio on the particular stimulation setting and activation status of the target T cell. We have identified a number of in vitro assays with response profiles that mimic features of dissociated cell populations from primary tumours thereby indicating these represent disease-relevant functional assays for the screening of immune checkpoint inhibitors in current and future development. Such in vitro assays may also support patient stratification of those likely to respond to immuno-oncology therapies in the wider population.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Wei SC, Duffy CR, Allison JP. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov. 2018;8:1069–1086. doi: 10.1158/2159-8290.CD-18-0367. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

