Orthogonal Bioluminescent Probes from Disubstituted Luciferins
- PMID: 33599497
- PMCID: PMC7955686
- DOI: 10.1021/acs.biochem.0c00894
Orthogonal Bioluminescent Probes from Disubstituted Luciferins
Abstract
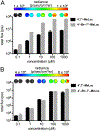
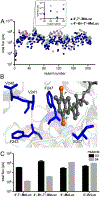
Bioluminescence imaging with luciferase-luciferin pairs is routinely used to monitor cellular functions. Multiple targets can be visualized in tandem using luciferases that process unique substrates, but only a handful of such orthogonal probes are known. Multiplexed studies require additional robust, light-emitting molecules. In this work, we report new luciferins for orthogonal imaging that comprise disubstituted cores. These probes were found to be bright emitters with various engineered luciferases. The unique patterns of light output also provided insight into enzyme-substrate interactions necessary for productive emission. Screening studies identified mutant luciferases that could preferentially process the disubstituted analogues, enabling orthogonal imaging with existing bioluminescent reporters. Further mutational analyses revealed the origins of substrate selectivity. Collectively, this work provides insights into luciferase-luciferin features relevant to bioluminescence and expands the number of probes for multicomponent tracking.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
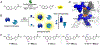
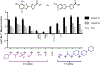




Similar articles
-
Building Biological Flashlights: Orthogonal Luciferases and Luciferins for in Vivo Imaging.Acc Chem Res. 2019 Nov 19;52(11):3039-3050. doi: 10.1021/acs.accounts.9b00391. Epub 2019 Oct 8. Acc Chem Res. 2019. PMID: 31593431 Free PMC article. Review.
-
Multicomponent Bioluminescence Imaging with Naphthylamino Luciferins.Chembiochem. 2021 Aug 17;22(16):2650-2654. doi: 10.1002/cbic.202100202. Epub 2021 Jun 30. Chembiochem. 2021. PMID: 34139065 Free PMC article.
-
Multicomponent Bioluminescence Imaging with a π-Extended Luciferin.J Am Chem Soc. 2020 Aug 19;142(33):14080-14089. doi: 10.1021/jacs.0c01064. Epub 2020 Aug 4. J Am Chem Soc. 2020. PMID: 32787261 Free PMC article.
-
Orthogonal Luciferase-Luciferin Pairs for Bioluminescence Imaging.J Am Chem Soc. 2017 Feb 15;139(6):2351-2358. doi: 10.1021/jacs.6b11737. Epub 2017 Feb 3. J Am Chem Soc. 2017. PMID: 28106389 Free PMC article.
-
Cage the firefly luciferin! - a strategy for developing bioluminescent probes.Chem Soc Rev. 2013 Jan 21;42(2):662-76. doi: 10.1039/c2cs35249d. Chem Soc Rev. 2013. PMID: 23099531 Review.
Cited by
-
Synthesis of 2-Cyanobenzothiazoles via Pd-Catalyzed/Cu-Assisted C-H Functionalization/Intramolecular C-S Bond Formation from N-Arylcyanothioformamides.Molecules. 2022 Dec 1;27(23):8426. doi: 10.3390/molecules27238426. Molecules. 2022. PMID: 36500519 Free PMC article.
-
Brightening up Biology: Advances in Luciferase Systems for in Vivo Imaging.ACS Chem Biol. 2021 Dec 17;16(12):2707-2718. doi: 10.1021/acschembio.1c00549. Epub 2021 Nov 15. ACS Chem Biol. 2021. PMID: 34780699 Free PMC article. Review.
-
Caged luciferins enable rapid multicomponent bioluminescence imaging.Photochem Photobiol. 2024 Jan-Feb;100(1):67-74. doi: 10.1111/php.13814. Epub 2023 May 31. Photochem Photobiol. 2024. PMID: 37259257 Free PMC article.
-
Biochemical Analysis Leads to Improved Orthogonal Bioluminescent Tools.Chembiochem. 2023 Mar 14;24(6):e202200726. doi: 10.1002/cbic.202200726. Epub 2023 Feb 10. Chembiochem. 2023. PMID: 36592373 Free PMC article.
-
Coumarin luciferins and mutant luciferases for robust multi-component bioluminescence imaging.Chem Sci. 2021 Aug 2;12(35):11684-11691. doi: 10.1039/d1sc03114g. eCollection 2021 Sep 15. Chem Sci. 2021. PMID: 34659703 Free PMC article.
References
-
- Prescher JA, Contag CH (2010) Guided by the light: visualizing biomolecular processes in living animals with bioluminescence, Curr. Opin. Chem. Biol 14, 80–89. - PubMed
-
- Contag CH, Spilman SD, Contag PR, Oshiro M, Eames B, Dennery P, Stevenson DK, Benaron DA . (1997) Visualizing gene expression in living mammals using a bioluminescent reporter, Photochem. Photobiol 66, 523–531. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

