Stringent thresholds in SARS-CoV-2 IgG assays lead to under-detection of mild infections
- PMID: 33602152
- PMCID: PMC7889711
- DOI: 10.1186/s12879-021-05878-2
Stringent thresholds in SARS-CoV-2 IgG assays lead to under-detection of mild infections
Abstract
Background: Thresholds for SARS-CoV-2 antibody assays have typically been determined using samples from symptomatic, often hospitalised, patients. In this setting the sensitivity and specificity of the best performing assays can both exceed 98%. However, antibody assay performance following mild infection is less clear.
Methods: We assessed quantitative IgG responses in a cohort of healthcare workers in Oxford, UK, with a high pre-test probability of Covid-19, in particular the 991/11,475(8.6%) who reported loss of smell/taste. We use anosmia/ageusia and other risk factors as probes for Covid-19 infection potentially undiagnosed by immunoassays by investigating their relationship with antibody readings either side of assay thresholds.
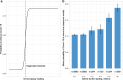
Results: The proportion of healthcare workers reporting anosmia/ageusia increased at antibody readings below diagnostic thresholds using an in-house ELISA (n = 9324) and the Abbott Architect chemiluminescent microparticle immunoassay (CMIA; n = 11,324): 426/906 (47%) reported anosmia/ageusia with a positive ELISA, 59/449 (13.1%) with high-negative and 326/7969 (4.1%) with low-negative readings. Similarly, by CMIA, 518/1093 (47.4%) with a positive result reported anosmia/ageusia, 106/686 (15.5%) with a high-negative and 358/9563 (3.7%) with a low-negative result. Adjusting for the proportion of staff reporting anosmia/ageusia suggests the sensitivity of both assays in mild infection is lower than previously reported: Oxford ELISA 89.8% (95%CI 86.6-92.8%) and Abbott CMIA 79.3% (75.9-82.7%).
Conclusion: Following mild SARS-CoV-2 infection 10-30% of individuals may have negative immunoassay results. While lowered diagnostic thresholds may result in unacceptable specificity, our findings have implications for epidemiological analyses and result interpretation in individuals with a high pre-test probability. Samples from mild PCR-confirmed infections should be included in SARS-CoV-2 immunoassay evaluations.
Keywords: Ageusia; Anosmia; Antibodies; COVID-19; SARS-CoV-2; Serology.
Conflict of interest statement
DWE declares lecture fees from Gilead, outside the submitted work. RJC is a founder shareholder and consultant to MIROBio, work outside the submitted work. No other author has a conflict of interest to declare.
Figures



References
-
- Office for National Statistics . Coronavirus (COVID-19) Infection Survey pilot: 2 July 2020. 2020.
-
- Lumley SF, O’Donnell D, Stoesser NE, et al. Antibodies to SARS-CoV-2 are associated with protection against reinfection. MedRxiv. 2020. 10.1101/2020.11.18.20234369.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

