IL-1 Inhibitors in the Treatment of Monogenic Periodic Fever Syndromes: From the Past to the Future Perspectives
- PMID: 33603750
- PMCID: PMC7884884
- DOI: 10.3389/fimmu.2020.619257
IL-1 Inhibitors in the Treatment of Monogenic Periodic Fever Syndromes: From the Past to the Future Perspectives
Abstract
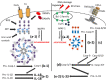
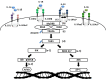
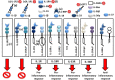
Autoinflammatory diseases (AIDs) represent a rare and heterogeneous group of disorders characterized by recurrent episodes of inflammation and a broad range of clinical manifestations. The most common symptoms involve recurrent fevers, musculoskeletal symptoms, and serositis; however, AIDs can also lead to life-threatening complications, such as macrophage activation syndrome (MAS) and systemic AA amyloidosis. Typical monogenic periodic fever syndromes include cryopyrin-associated periodic fever syndrome (CAPS), tumor necrosis factor receptor-associated periodic syndrome (TRAPS), mevalonate kinase deficiency/hyper IgD syndrome (MKD/HIDS), and familial Mediterranean fever (FMF). However, a number of other clinical entities, such as systemic juvenile idiopathic arthritis (sJIA), adult-onset Still's disease (AOSD), Kawasaki disease (KD) and idiopathic recurrent pericarditis (IRP), display similar phenotypical and immunological features to AIDs. All these diseases are pathophysiologicaly characterized by dysregulation of the innate immune system and the central pathogenic role is attributed to the IL-1 cytokine family (IL-1α, IL-1β, IL-1Ra, IL-18, IL-36Ra, IL-36α, IL-37, IL-36β, IL-36g, IL-38, and IL-33). Therefore, reasonable therapeutic approaches aim to inhibit these cytokines and their pathways. To date, several anti-IL-1 therapies have evolved. Each drug differs in structure, mechanism of action, efficacy for the treatment of selected diseases, and side effects. Most of the available data regarding the efficacy and safety of IL-1 inhibitors are related to anakinra, canakinumab, and rilonacept. Other promising therapeutics, such as gevokizumab, tadekinig alfa, and tranilast are currently undergoing clinical trials. In this review, we provide sophisticated and up-to-date insight into the therapeutic uses of different IL-1 inhibitors in monogenic periodic fever syndromes.
Keywords: CAPS; FMF; IL-1; TRAPS; anakinra; canakinumab; rilonacept.
Copyright © 2021 Malcova, Strizova, Milota, Striz, Sediva, Cebecauerova and Horvath.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

