Jasmonic acid biosynthesis by fungi: derivatives, first evidence on biochemical pathways and culture conditions for production
- PMID: 33604199
- PMCID: PMC7869668
- DOI: 10.7717/peerj.10873
Jasmonic acid biosynthesis by fungi: derivatives, first evidence on biochemical pathways and culture conditions for production
Abstract
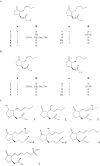
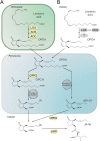
Jasmonic acid (JA) and its derivatives called jasmonates (JAs) are lipid-derived signalling molecules that are produced by plants and certain fungi. Beside this function, JAs have a great variety of applications in flavours and fragrances production. In addition, they may have a high potential in agriculture. JAs protect plants against infections. Although there is much information on the biosynthesis and function of JA concerning plants, knowledge on these aspects is still scarce for fungi. Taking into account the practical importance of JAs, the objective of this review is to summarize knowledge on the occurrence of JAs from fungal culture media, their biosynthetic pathways and the culture conditions for optimal JA production as an alternative source for the production of these valuable metabolites.
Keywords: Culture medium; Fungi; Jasmonic acid; Metabolic pathway; Oxylipin; Submerged fermentation.
© 2021 Eng et al.
Conflict of interest statement
Ivo Feussner is an Academic Editor for PeerJ.
Figures
References
-
- Aldridge DC, Galt S, Giles D, Turner WB. Metabolites of Lasiodiplodia theobromae. Journal of the Chemical Society C: Organic. 1971;1971:1623–1627. doi: 10.1039/j39710001623. - DOI
-
- Almeida G, Altuna B, Michelena G. Espoluración en Botryodiplodia theobromae—influencia de la luz en la producción de picnidios. Laboract Acta. 2001;13:89–92.
-
- Almeida G, Klibansky M, Altuna B, Eng F, Legrá S, Armenteros S. Some considerations of the carbon sources used in jasmonic acid production. Revista Iberoamericana de Micología. 1999;16:146–148. - PubMed
-
- Altuna B, Klibansky M, Almeida G, Eng F, Legra S, Armenteros S. Jasmonic acid detection as a new plant growth regulator in strains of Botryodiplodia theobromae. Revista Sobre los Derivados de la Cana de Azucar. 1996;30:16–21.
LinkOut - more resources
Full Text Sources
Other Literature Sources

