Retinoic acid signaling restricts the size of the first heart field within the anterior lateral plate mesoderm
- PMID: 33607112
- PMCID: PMC7987825
- DOI: 10.1016/j.ydbio.2021.02.005
Retinoic acid signaling restricts the size of the first heart field within the anterior lateral plate mesoderm
Abstract
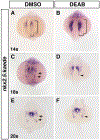
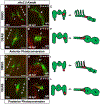
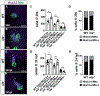
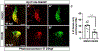
Retinoic acid (RA) signaling is required to restrict heart size through limiting the posterior boundary of the vertebrate cardiac progenitor field within the anterior lateral plate mesoderm (ALPM). However, we still do not fully understand how different cardiac progenitor populations that contribute to the developing heart, including earlier-differentiating first heart field (FHF), later-differentiating second heart field (SHF), and neural crest-derived progenitors, are each affected in RA-deficient embryos. Here, we quantified the number of cardiac progenitors and differentiating cardiomyocytes (CMs) in RA-deficient zebrafish embryos. While Nkx2.5+ cells were increased overall in the nascent hearts of RA-deficient embryos, unexpectedly, we found that the major effect within this population was a significant expansion in the number of differentiating FHF CMs. In contrast to the expansion of the FHF, there was a progressive decrease in SHF progenitors at the arterial pole as the heart tube elongated. Temporal differentiation assays and immunostaining in RA-deficient embryos showed that the outflow tracts (OFTs) of the hearts were significantly smaller, containing fewer differentiated SHF-derived ventricular CMs and a complete absence of SHF-derived smooth muscle at later stages. At the venous pole of the heart, pacemaker cells of the sinoatrial node also failed to differentiate in RA-deficient embryos. Interestingly, genetic lineage tracing showed that the number of neural-crest derived CMs was not altered within the enlarged hearts of RA-deficient zebrafish embryos. Altogether, our data show that the enlarged hearts in RA-deficient zebrafish embryos are comprised of an expansion in earlier differentiating FHF-derived CMs coupled with a progressive depletion of the SHF, suggesting RA signaling determines the relative ratios of earlier- and later-differentiation cardiac progenitors within an expanded cardiac progenitor pool.
Keywords: Atrium; Cardiomyocyte progenitors; First heart field; Heart development; Outflow tract; Retinoic acid signaling; Second heart field; Sinoatrial node; Ventricle; Zebrafish.
Copyright © 2021 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare no competing interests.
Figures






References
-
- Begemann G, Schilling TF, Rauch GJ, Geisler R, Ingham PW, 2001. The zebrafish neckless mutation reveals a requirement for raldh2 in mesodermal signals that pattern the hindbrain. Development 128, 3081–3094. - PubMed
-
- Buckingham M, Meilhac S, Zaffran S, 2005. Building the mammalian heart from two sources of myocardial cells. Nat. Rev. Genet 6, 826–835. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

