Development of a novel immune-related lncRNA signature as a prognostic classifier for endometrial carcinoma
- PMID: 33613104
- PMCID: PMC7893582
- DOI: 10.7150/ijbs.51207
Development of a novel immune-related lncRNA signature as a prognostic classifier for endometrial carcinoma
Abstract
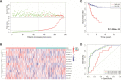
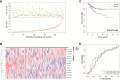
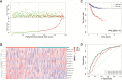
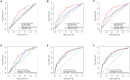
Endometrial carcinoma (EnCa) is one of the deadliest gynecological malignancies. The purpose of the current study was to develop an immune-related lncRNA prognostic signature for EnCa. In the current research, a series of systematic bioinformatics analyses were conducted to develop a novel immune-related lncRNA prognostic signature to predict disease-free survival (DFS) and response to immunotherapy and chemotherapy in EnCa. Based on the newly developed signature, immune status and mutational loading between high‑ and low‑risk groups were also compared. A novel 13-lncRNA signature associated with DFS of EnCa patients was ultimately developed using systematic bioinformatics analyses. The prognostic signature allowed us to distinguish samples with different risks with relatively high accuracy. In addition, univariate and multivariate Cox regression analyses confirmed that the signature was an independent factor for predicting DFS in EnCa. Moreover, a predictive nomogram combined with the risk signature and clinical stage was constructed to accurately predict 1-, 2-, 3-, and 5-year DFS of EnCa patients. Additionally, EnCa patients with different levels of risk had markedly different immune statuses and mutational loadings. Our findings indicate that the immune-related 13-lncRNA signature is a promising classifier for prognosis and response to immunotherapy and chemotherapy for EnCa.
Keywords: bioinformatics; endometrial carcinoma; immune-related lncRNA; signature.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures



Similar articles
-
Identifying immune subtypes of uterine corpus endometrial carcinoma and a four-paired-lncRNA signature with immune-related lncRNAs.Exp Biol Med (Maywood). 2022 Feb;247(3):221-236. doi: 10.1177/15353702211053588. Epub 2021 Oct 27. Exp Biol Med (Maywood). 2022. PMID: 34704492 Free PMC article.
-
Identification of an Immune-Related LncRNA Prognostic Signature in Uterine Corpus Endometrial Carcinoma Patients.Clin Lab. 2021 Nov 1;67(11). doi: 10.7754/Clin.Lab.2021.210401. Clin Lab. 2021. PMID: 34758230
-
A novel lncRNA-focus expression signature for survival prediction in endometrial carcinoma.BMC Cancer. 2018 Jan 5;18(1):39. doi: 10.1186/s12885-017-3983-0. BMC Cancer. 2018. PMID: 29304762 Free PMC article.
-
High-Grade Endometrial Cancer: Molecular Subtypes, Current Challenges, and Treatment Options.Reprod Sci. 2024 Sep;31(9):2541-2559. doi: 10.1007/s43032-024-01544-5. Epub 2024 Apr 24. Reprod Sci. 2024. PMID: 38658487 Review.
-
Immunomodulation in endometrial cancer.Int J Gynecol Cancer. 2009 May;19(4):734-40. doi: 10.1111/IGC.0b013e3181a12f7f. Int J Gynecol Cancer. 2009. PMID: 19509580 Review.
Cited by
-
Development and validation of a cancer-associated fibroblast-derived lncRNA signature for predicting clinical outcomes in colorectal cancer.Front Immunol. 2022 Jul 29;13:934221. doi: 10.3389/fimmu.2022.934221. eCollection 2022. Front Immunol. 2022. PMID: 35967425 Free PMC article.
-
Integrated analysis on the N6-methyladenosine-related long noncoding RNAs prognostic signature, immune checkpoints, and immune cell infiltration in clear cell renal cell carcinoma.Immun Inflamm Dis. 2021 Dec;9(4):1596-1612. doi: 10.1002/iid3.513. Epub 2021 Aug 25. Immun Inflamm Dis. 2021. PMID: 34432955 Free PMC article.
-
A new marker constructed from immune-related lncRNA pairs can be used to predict clinical treatment effects and prognosis: in-depth exploration of underlying mechanisms in HNSCC.World J Surg Oncol. 2023 Aug 17;21(1):250. doi: 10.1186/s12957-023-03066-x. World J Surg Oncol. 2023. PMID: 37592311 Free PMC article.
-
The Long Noncoding Transcript HNSCAT1 Activates KRT80 and Triggers Therapeutic Efficacy in Head and Neck Squamous Cell Carcinoma.Oxid Med Cell Longev. 2022 Aug 4;2022:4156966. doi: 10.1155/2022/4156966. eCollection 2022. Oxid Med Cell Longev. 2022. PMID: 35965679 Free PMC article.
-
Necrotic related-lncRNAs: Prediction of prognosis and differentiation between cold and hot tumors in head and neck squamous cell carcinoma.Medicine (Baltimore). 2023 Jun 9;102(23):e33994. doi: 10.1097/MD.0000000000033994. Medicine (Baltimore). 2023. PMID: 37335630 Free PMC article.
References
-
- Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017;67:7–30. - PubMed
-
- Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M. et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–86. - PubMed
-
- Mang C, Birkenmaier A, Cathomas G, Humburg J. Endometrioid endometrial adenocarcinoma: an increase of G3 cancers? Arch Gynecol Obstet. 2017;295:1435–40. - PubMed
-
- Green AK, Feinberg J, Makker V. A Review of Immune Checkpoint Blockade Therapy in Endometrial Cancer. Am Soc Clin Oncol Educ Book. 2020;40:1–7. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

