A Case of Lung Adenocarcinoma with Long-Term Response after Late-Onset Pembrolizumab-Induced Acute Adrenal Insufficiency
- PMID: 33613234
- PMCID: PMC7879310
- DOI: 10.1159/000508068
A Case of Lung Adenocarcinoma with Long-Term Response after Late-Onset Pembrolizumab-Induced Acute Adrenal Insufficiency
Abstract
Pembrolizumab is an anti-programmed cell death protein-1 antibody that is mainly used for the treatment of non-small cell lung cancer (NSCLC). Immune-related adverse events can be caused by immune checkpoint inhibitors; however, few case reports evaluate the prognosis of patients with NSCLC with late-onset immune-related adverse events. In this case, a 63-year-old man with stage IVA lung adenocarcinoma received pembrolizumab as first-line therapy and achieved a complete response. The patient developed hypothyroidism and skin toxicity owing to pembrolizumab over the course of treatment; however, the patient continued with pembrolizumab. The patient discontinued pembrolizumab after 20 cycles owing to appetite loss from 14 months after the initiation of pembrolizumab. Two months later, the symptoms worsened and the patient was taken to hospital by an ambulance owing to movement difficulty. The patient was diagnosed with acute adrenal insufficiency by endocrinological examinations. The condition of the patient improved after hydrocortisone treatment. Sixteen months have passed without the readministration of pembrolizumab and no recurrence of lung adenocarcinoma has been observed. Late-onset, severe, and diverse immune-related adverse events may be a favorable prognostic factor associated with survival.
Keywords: Adrenal insufficiency; Immune-related adverse events; Lung adenocarcinoma; Pembrolizumab; Prognostic factor.
Copyright © 2021 by S. Karger AG, Basel.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures
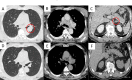
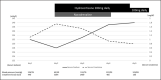
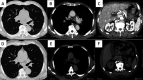
Similar articles
-
Simultaneous Presentation of Secondary Adrenal Insufficiency and Primary Hypothyroidism due to Pembrolizumab: A Case Report.J Investig Med High Impact Case Rep. 2023 Jan-Dec;11:23247096231194401. doi: 10.1177/23247096231194401. J Investig Med High Impact Case Rep. 2023. PMID: 37642143 Free PMC article.
-
Late-onset programmed cell death protein-1 inhibitor-induced pneumonitis after cessation of nivolumab or pembrolizumab in patients with advanced non-small cell lung cancer: a case series.Transl Lung Cancer Res. 2021 Mar;10(3):1576-1581. doi: 10.21037/tlcr-20-582. Transl Lung Cancer Res. 2021. PMID: 33889531 Free PMC article.
-
Intralymphatic histiocytosis in a patient with lung adenocarcinoma treated with pembrolizumab: a case report.J Immunother Cancer. 2019 Feb 27;7(1):59. doi: 10.1186/s40425-019-0534-z. J Immunother Cancer. 2019. PMID: 30813943 Free PMC article.
-
Isolated adrenocorticotropic hormone deficiency and thyroiditis associated with nivolumab therapy in a patient with advanced lung adenocarcinoma: a case report and review of the literature.J Med Case Rep. 2019 Mar 26;13(1):88. doi: 10.1186/s13256-019-2002-2. J Med Case Rep. 2019. PMID: 30909965 Free PMC article. Review.
-
Pericardial tamponade during pembrolizumab treatment in a patient with advanced lung adenocarcinoma: A case report and review of the literature.Thorac Cancer. 2020 May;11(5):1350-1353. doi: 10.1111/1759-7714.13399. Epub 2020 Mar 17. Thorac Cancer. 2020. PMID: 32181993 Free PMC article. Review.
Cited by
-
Late-onset acute type 1 diabetes mellitus 7 months after discontinuation of pembrolizumab against lung cancer.Thorac Cancer. 2023 Jan;14(1):81-84. doi: 10.1111/1759-7714.14736. Epub 2022 Nov 21. Thorac Cancer. 2023. PMID: 36411591 Free PMC article.
-
Isolated Adrenocorticotropic Hormone Deficiency Associated with Severe Hyperkalemia During Pembrolizumab Therapy in a Patient with Ureteral Cancer and an Ileal Conduit: A Case Report and Literature Review.Am J Case Rep. 2021 Jul 15;22:e931639. doi: 10.12659/AJCR.931639. Am J Case Rep. 2021. PMID: 34262010 Free PMC article. Review.
-
Pembrolizumab-Induced Adrenal Insufficiency in Patients with Untreated Advanced Non-Small Cell Lung Cancer: A Case Series.Case Rep Oncol. 2021 Nov 5;14(3):1561-1566. doi: 10.1159/000519597. eCollection 2021 Sep-Dec. Case Rep Oncol. 2021. PMID: 34949996 Free PMC article.
-
Clinical course and management of insidious adrenal crisis manifested initially as hyperpyrexia secondary to pembrolizumab: Case reports and literature review.Front Oncol. 2022 Aug 11;12:981084. doi: 10.3389/fonc.2022.981084. eCollection 2022. Front Oncol. 2022. PMID: 36033475 Free PMC article.
-
Pembrolizumab-induced secondary adrenal insufficiency due to adrenocorticotrophic hormone deficiency in a patient with non-small-cell lung carcinoma: a case report.J Pharm Health Care Sci. 2024 Feb 16;10(1):10. doi: 10.1186/s40780-024-00332-2. J Pharm Health Care Sci. 2024. PMID: 38365819 Free PMC article.
References
-
- Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016 Nov;375((19)):1823–33. - PubMed
-
- Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016 Apr;387((10027)):1540–50. - PubMed
-
- Mok TSK, Wu YL, Kudaba I, Kowalski DM, Cho BC, Turna HZ, et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet. 2019 May;393((10183)):1819–30. - PubMed
-
- Spain L, Diem S, Larkin J. Management of toxicities of immune checkpoint inhibitors. Cancer Treat Rev. 2016 Mar;44:51–60. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

