Gray Matter Deterioration Pattern During Alzheimer's Disease Progression: A Regions-of-Interest Based Surface Morphometry Study
- PMID: 33613265
- PMCID: PMC7886803
- DOI: 10.3389/fnagi.2021.593898
Gray Matter Deterioration Pattern During Alzheimer's Disease Progression: A Regions-of-Interest Based Surface Morphometry Study
Abstract
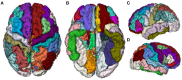
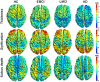
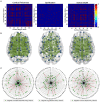
Accurate detection of the regions of Alzheimer's disease (AD) lesions is critical for early intervention to effectively slow down the progression of the disease. Although gray matter volumetric abnormalities are commonly detected in patients with mild cognition impairment (MCI) and patients with AD, the gray matter surface-based deterioration pattern associated with the progression of the disease from MCI to AD stages is largely unknown. To identify group differences in gray matter surface morphometry, including cortical thickness, the gyrification index (GI), and the sulcus depth, 80 subjects from the Alzheimer's Disease Neuroimaging Initiative (ADNI) database were split into healthy controls (HCs; N = 20), early MCIs (EMCI; N = 20), late MCIs (LMCI; N = 20), and ADs (N = 20). Regions-of-interest (ROI)-based surface morphometry was subsequently studied and compared across the four stage groups to characterize the gray matter deterioration during AD progression. Co-alteration patterns (Spearman's correlation coefficient) across the whole brain were also examined. Results showed that patients with MCI and AD exhibited a significant reduction in cortical thickness (p < 0.001) mainly in the cingulate region (four subregions) and in the temporal (thirteen subregions), parietal (five subregions), and frontal (six subregions) lobes compared to HCs. The sulcus depth of the eight temporal, four frontal, four occipital, and eight parietal subregions were also significantly affected (p < 0.001) by the progression of AD. The GI was shown to be insensitive to AD progression (only three subregions were detected with a significant difference, p < 0.001). Moreover, Spearman's correlation analysis confirmed that the co-alteration pattern of the cortical thickness and sulcus depth indices is predominant during AD progression. The findings highlight the relevance between gray matter surface morphometry and the stages of AD, laying the foundation for in vivo tracking of AD progression. The co-alteration pattern of surface-based morphometry would improve the researchers' knowledge of the underlying pathologic mechanisms in AD.
Keywords: Alzheimer's disease; cognition impairment; gray matter; magnetic resonance imaging; surface morphometry.
Copyright © 2021 Wu, Peng, Hong and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Patterns of Grey Matter Atrophy at Different Stages of Parkinson's and Alzheimer's Diseases and Relation to Cognition.Brain Topogr. 2019 Jan;32(1):142-160. doi: 10.1007/s10548-018-0675-2. Epub 2018 Sep 11. Brain Topogr. 2019. PMID: 30206799
-
The Structural Changes of Frontal Subregions and Their Correlations with Cognitive Impairment in Patients with Alzheimer's Disease.J Integr Neurosci. 2023 Jul 20;22(4):99. doi: 10.31083/j.jin2204099. J Integr Neurosci. 2023. PMID: 37519164
-
Differences in Cerebral Structure Associated With Depressive Symptoms in the Elderly With Alzheimer's Disease.Front Aging Neurosci. 2020 May 12;12:107. doi: 10.3389/fnagi.2020.00107. eCollection 2020. Front Aging Neurosci. 2020. PMID: 32477094 Free PMC article.
-
Voxel-based morphometry in Alzheimer's disease.Expert Rev Neurother. 2008 Nov;8(11):1691-702. doi: 10.1586/14737175.8.11.1691. Expert Rev Neurother. 2008. PMID: 18986240 Review.
-
Brain-printing biometrics underlying mechanism as an early diagnostic technique for Alzheimer's disease neurodegenerative type.Curr Res Physiol. 2021 Oct 2;4:216-222. doi: 10.1016/j.crphys.2021.09.005. eCollection 2021. Curr Res Physiol. 2021. PMID: 34746841 Free PMC article. Review.
Cited by
-
Revealing the correlations between brain cortical characteristics and susceptibility genes for Alzheimer disease: a cross-sectional study.Quant Imaging Med Surg. 2023 Apr 1;13(4):2451-2465. doi: 10.21037/qims-22-602. Epub 2023 Mar 22. Quant Imaging Med Surg. 2023. PMID: 37064375 Free PMC article.
-
Progranulin and neuropathological features of Alzheimer's disease: longitudinal study.Aging Clin Exp Res. 2024 Mar 5;36(1):55. doi: 10.1007/s40520-024-02715-9. Aging Clin Exp Res. 2024. PMID: 38441695 Free PMC article.
-
Extra cup of tea intake associated with increased risk of Alzheimer's disease: Genetic insights from Mendelian randomization.Front Nutr. 2023 Jan 25;10:1052281. doi: 10.3389/fnut.2023.1052281. eCollection 2023. Front Nutr. 2023. PMID: 36761219 Free PMC article.
-
Adversarial Learning for MRI Reconstruction and Classification of Cognitively Impaired Individuals.IEEE Access. 2024;12:83169-83182. doi: 10.1109/access.2024.3408840. Epub 2024 Jun 3. IEEE Access. 2024. PMID: 39148927 Free PMC article.
-
Automated High-Definition MRI Processing Routine Robustly Detects Longitudinal Morphometry Changes in Alzheimer's Disease Patients.Front Aging Neurosci. 2022 Jun 7;14:832828. doi: 10.3389/fnagi.2022.832828. eCollection 2022. Front Aging Neurosci. 2022. PMID: 35747446 Free PMC article.
References
-
- Beejesh A. G., Gopi V. P., Hemanth J. (2019). Brain MR kurtosis imaging study: contrasting gray and white matter. Cogn. Syst. Res. 55, 135–145. 10.1016/j.cogsys.2019.01.005 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

