Unraveling GLUT-mediated transcytosis pathway of glycosylated nanodisks
- PMID: 33613735
- PMCID: PMC7878461
- DOI: 10.1016/j.ajps.2020.07.001
Unraveling GLUT-mediated transcytosis pathway of glycosylated nanodisks
Abstract
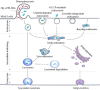
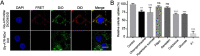
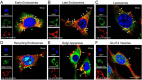
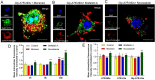
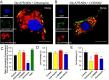
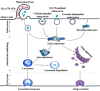
Glucose transporter (GLUT)-mediated transcytosis has been validated as an efficient method to cross the blood-brain barrier and enhance brain transport of nanomedicines. However, the transcytosis process remains elusive. Glycopeptide-modified nanodisks (Gly-A7R-NDs), which demonstrated high capacity of brain targeting via GLUT-mediated transcytosis in our previous reports, were utilized to better understand the whole transcytosis process. Gly-A7R-NDs internalized brain capillary endothelial cells mainly via GLUT-mediated/clathrin dependent endocytosis and macropinocytosis. The intracellular Gly-A7R-NDs remained intact, and the main excretion route of Gly-A7R-NDs was lysosomal exocytosis. Glycosylation of nanomedicine was crucial in GLUT-mediated transcytosis, while morphology did not affect the efficiency. This study highlights the pivotal roles of lysosomal exocytosis in the process of GLUT-mediated transcytosis, providing a new impetus to development of brain targeting drug delivery by accelerating lysosomal exocytosis.
Keywords: Blood-brain barrier; Glucose transporter; Glycosylation; Lysosomal exocytosis; Transcytosis.
© 2020 Shenyang Pharmaceutical University. Published by Elsevier B.V.
Conflict of interest statement
None.
Figures

Similar articles
-
Clathrin-dependent entry and vesicle-mediated exocytosis define insulin transcytosis across microvascular endothelial cells.Mol Biol Cell. 2015 Feb 15;26(4):740-50. doi: 10.1091/mbc.E14-08-1307. Epub 2014 Dec 24. Mol Biol Cell. 2015. PMID: 25540431 Free PMC article.
-
Nanodisk-based glioma-targeted drug delivery enabled by a stable glycopeptide.J Control Release. 2018 Aug 28;284:26-38. doi: 10.1016/j.jconrel.2018.06.006. Epub 2018 Jun 7. J Control Release. 2018. PMID: 29885416
-
The transport mechanisms of polymer nanoparticles in Caco-2 epithelial cells.Biomaterials. 2013 Aug;34(25):6082-98. doi: 10.1016/j.biomaterials.2013.04.053. Epub 2013 May 18. Biomaterials. 2013. PMID: 23694903
-
Transcytosis of macromolecules at the blood-brain barrier.Adv Pharmacol. 2014;71:147-63. doi: 10.1016/bs.apha.2014.06.001. Epub 2014 Aug 22. Adv Pharmacol. 2014. PMID: 25307216 Review.
-
The Role of Caveolae on Albumin Passage through Glomerular Endothelial and Epithelial Cells: The New Etiology of Urinary Albumin Excretion.Contrib Nephrol. 2018;195:1-11. doi: 10.1159/000486929. Epub 2018 May 7. Contrib Nephrol. 2018. PMID: 29734145 Review.
Cited by
-
All-stage targeted nanodiscs for glioma treatment by inducing cuproptosis and apoptosis of cancer cells and cancer stem cells.Asian J Pharm Sci. 2025 Apr;20(2):101010. doi: 10.1016/j.ajps.2024.101010. Epub 2024 Dec 17. Asian J Pharm Sci. 2025. PMID: 40182135 Free PMC article.
-
Impact of Green Gold Nanoparticle Coating on Internalization, Trafficking, and Efficiency for Photothermal Therapy of Skin Cancer.ACS Omega. 2023 Jan 20;8(4):4092-4105. doi: 10.1021/acsomega.2c07054. eCollection 2023 Jan 31. ACS Omega. 2023. PMID: 36743010 Free PMC article.
-
Crossing the Blood-Brain Barrier: Innovations in Receptor- and Transporter-Mediated Transcytosis Strategies.Pharmaceutics. 2025 May 28;17(6):706. doi: 10.3390/pharmaceutics17060706. Pharmaceutics. 2025. PMID: 40574019 Free PMC article. Review.
-
Exocytosis of Nanoparticles: A Comprehensive Review.Nanomaterials (Basel). 2023 Jul 30;13(15):2215. doi: 10.3390/nano13152215. Nanomaterials (Basel). 2023. PMID: 37570533 Free PMC article. Review.
-
BRD4 Degradation Enhanced Glioma Sensitivity to Temozolomide by Regulating Notch1 via Glu-Modified GSH-Responsive Nanoparticles.Adv Sci (Weinh). 2024 Dec;11(48):e2409753. doi: 10.1002/advs.202409753. Epub 2024 Nov 15. Adv Sci (Weinh). 2024. PMID: 39544152 Free PMC article.
References
-
- Agrahari V., Burnouf P., Burnouf T., Agrahari V. Nanoformulation properties, characterization, and behavior in complex biological matrices: challenges and opportunities for brain-targeted drug delivery applications and enhanced translational potential. Adv Drug Deliver Rev. 2019;148:146–180. - PubMed
-
- Pandit R., Chen L., Götz J. The blood-brain barrier: physiology and strategies for drug delivery. Adv Drug Deliver Rev. 2019:S169–S409. - PubMed
-
- Deng D., Xu C., Sun P., Wu J., Yan C., Hu M. Crystal structure of the human glucose transporter GLUT1. Nature. 2014;510(7503):121–125. - PubMed
-
- Shao K., Ding N., Huang S., Ren S., Zhang Y., Kuang Y. Smart nanodevice combined tumor-specific vector with cellular microenvironment-triggered property for highly effective antiglioma therapy. ACS Nano. 2014;8(2):1191–1203. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

