Branched amphotericin functional poly(N- iso propyl acrylamide): an antifungal polymer
- PMID: 33614095
- PMCID: PMC7890487
- DOI: 10.1098/rsos.201655
Branched amphotericin functional poly(N- iso propyl acrylamide): an antifungal polymer
Abstract
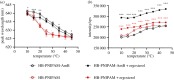
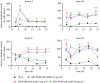
Branched poly(N-isopropylacrylamide) was functionalized with Amphotericin B (AmB) at the chain ends to produce an antifungal material. The polymer showed antifungal properties against AmB-sensitive strains of Candida albicans, Fusarium keratoplasticum and Aspergillus flavus (minimal inhibitory concentration ranged from 5 to 500 µg ml-1) but was not effective against an AmB resistant strain of C. albicans nor against Candida tropicalis. The polymer end groups bound to the AmB target, ergosterol, and the fluorescence spectrum of a dye used as a solvatochromic probe, Nile red, was blue shifted indicating that segments of the polymer became desolvated on binding. The polymer was less toxic to corneal and renal epithelial cells and explanted corneal tissue than the free drug. Also, the polymer did not induce reactive oxygen species release from peripheral blood mononuclear cells, nor did it cause a substantial release of the proinflammatory cytokines, tumour necrosis factor-α and interleukin-1β (at 0.5 mg ml-1).
Keywords: branched polymer; fungi; smart polymers.
© 2021 The Authors.
Conflict of interest statement
There are no conflicting financial interests of the authors.
Figures





References
-
- Yuan SJ, Xu FJ, Pehkonen SO, Ting YP, Neoh KG, Kang ET. 2009. Grafting of antibacterial polymers on stainless steel via surface-initiated atom transfer radical polymerization for inhibiting biocorrosion by Desulfovibrio desulfuricans. Biotechnol. Bioeng. 103, 268–281. (10.1002/bit.22252) - DOI - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
