Consistent Long-Term Therapeutic Efficacy of Human Umbilical Cord Matrix-Derived Mesenchymal Stromal Cells After Myocardial Infarction Despite Individual Differences and Transient Engraftment
- PMID: 33614654
- PMCID: PMC7890004
- DOI: 10.3389/fcell.2021.624601
Consistent Long-Term Therapeutic Efficacy of Human Umbilical Cord Matrix-Derived Mesenchymal Stromal Cells After Myocardial Infarction Despite Individual Differences and Transient Engraftment
Erratum in
-
Corrigendum: Consistent long-term therapeutic efficacy of human umbilical cord matrix-derived mesenchymal stromal cells after myocardial infarction despite individual differences and transient engraftment.Front Cell Dev Biol. 2023 Aug 18;11:1265005. doi: 10.3389/fcell.2023.1265005. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37664464 Free PMC article.
Abstract
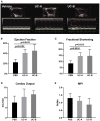
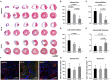
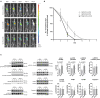
Human mesenchymal stem cells gather special interest as a universal and feasible add-on therapy for myocardial infarction (MI). In particular, human umbilical cord matrix-derived mesenchymal stromal cells (UCM-MSC) are advantageous since can be easily obtained and display high expansion potential. Using isolation protocols compliant with cell therapy, we previously showed UCM-MSC preserved cardiac function and attenuated remodeling 2 weeks after MI. In this study, UCM-MSC from two umbilical cords, UC-A and UC-B, were transplanted in a murine MI model to investigate consistency and durability of the therapeutic benefits. Both cellular products improved cardiac function and limited adverse cardiac remodeling 12 weeks post-ischemic injury, supporting sustained and long-term beneficial therapeutic effect. Donor associated variability was found in the modulation of cardiac remodeling and activation of the Akt-mTOR-GSK3β survival pathway. In vitro, the two cell products displayed similar ability to induce the formation of vessel-like structures and comparable transcriptome in normoxia and hypoxia, apart from UCM-MSCs proliferation and expression differences in a small subset of genes associated with MHC Class I. These findings support that UCM-MSC are strong candidates to assist the treatment of MI whilst calling for the discussion on methodologies to characterize and select best performing UCM-MSC before clinical application.
Keywords: Wharton's jelly; cardiac fibrosis; cell therapy; donor variability; mesenchymal stromal (or stem) cells; myocardial infarction; regeneration/repair; umbilical cord matrix derived mesenchymal stromal cells (hUCM-MSCs).
Copyright © 2021 Laundos, Vasques-Nóvoa, Gomes, Sampaio-Pinto, Cruz, Cruz, Santos, Barcia, Pinto-do-Ó and Nascimento.
Conflict of interest statement
HC and PC were shareholders of ECBio S.A. JS and RB were employees of ECBio S.A. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Arslan F., Lai R. C., Smeets M. B., Akeroyd L., Choo A., Aguor E. N., et al. (2013). Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/Akt pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res. 10, 301–312. 10.1016/j.scr.2013.01.002 - DOI - PubMed
-
- Bartolucci J., Verdugo F. J., González P. L., Larrea R. E., Abarzua E., Goset C., et al. (2017). Safety and efficacy of the intravenous infusion of umbilical cord mesenchymal stem cells in patients with heart failure: a phase 1/2 randomized controlled trial (rimecard trial [randomized clinical trial of intravenous infusion umbilical cord mesenchymal stem cells on cardiopathy]). Circ. Res. 121, 1192–1204. 10.1161/circresaha.117.310712 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

