Effects of colon-targeted vitamins on the composition and metabolic activity of the human gut microbiome- a pilot study
- PMID: 33615992
- PMCID: PMC7899684
- DOI: 10.1080/19490976.2021.1875774
Effects of colon-targeted vitamins on the composition and metabolic activity of the human gut microbiome- a pilot study
Abstract
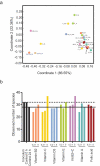
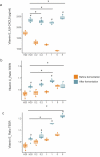
An increasing body of evidence has shown that gut microbiota imbalances are linked to diseases. Currently, the possibility of regulating gut microbiota to reverse these perturbations by developing novel therapeutic and preventive strategies is being extensively investigated. The modulatory effect of vitamins on the gut microbiome and related host health benefits remain largely unclear. We investigated the effects of colon-delivered vitamins A, B2, C, D, and E on the gut microbiota using a human clinical study and batch fermentation experiments, in combination with cell models for the assessment of barrier and immune functions. Vitamins C, B2, and D may modulate the human gut microbiome in terms of metabolic activity and bacterial composition. The most distinct effect was that of vitamin C, which significantly increased microbial alpha diversity and fecal short-chain fatty acids compared to the placebo. The remaining vitamins tested showed similar effects on microbial diversity, composition, and/or metabolic activity in vitro, but in varying degrees. Here, we showed that vitamins may modulate the human gut microbiome. Follow-up studies investigating targeted delivery of vitamins to the colon may help clarify the clinical significance of this novel concept for treating and preventing dysbiotic microbiota-related human diseases. Trial registration: ClinicalTrials.gov, NCT03668964. Registered 13 September 2018 - Retrospectively registered, https://clinicaltrials.gov/ct2/show/NCT03668964.
Keywords: Vitamins; dysbiosis; gut microbiome; targeted delivery.
Figures



References
-
- Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, Scott K, Stanton C, Swanson KS, Cani PD, et al. 2017. Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 14(8):491–502. doi: 10.1038/nrgastro.2017.75. - DOI - PubMed
-
- von Martels JZH, Bourgonje AR, Klaassen MAY, Alkhalifah HAA, Sadaghian Sadabad M, Vich Vila A, Gacesa R, Gabriëls RY, Steinert RE, Jansen BH, et al. Riboflavin supplementation in patients with Crohn’s disease (RISE-UP study). J Crohns Colitis. 2019;14(5):595–607. doi: 10.1093/ecco-jcc/jjz208. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
