A Role for Auxin in Triggering Lamina Outgrowth of Unifacial Leaves
- PMID: 33620494
- PMCID: PMC8195525
- DOI: 10.1093/plphys/kiab087
A Role for Auxin in Triggering Lamina Outgrowth of Unifacial Leaves
Abstract
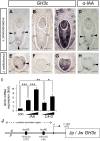
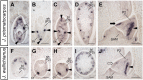
A common morphological feature of typical angiosperms is the patterning of lateral organs along primary axes of asymmetry-a proximodistal, a mediolateral, and an adaxial-abaxial axis. Angiosperm leaves usually have distinct adaxial-abaxial identity, which is required for the development of a flat shape. By contrast, many unifacial leaves, consisting of only the abaxial side, show a flattened morphology. This implicates a unique mechanism that allows leaf flattening independent of adaxial-abaxial identity. In this study, we report a role for auxin in outgrowth of unifacial leaves. In two closely related unifacial-leaved species of Juncaceae, Juncus prismatocarpus with flattened leaves, and Juncus wallichianus with transversally radialized leaves, the auxin-responsive gene GLYCOSIDE HYDROLASE3 displayed spatially different expression patterns within leaf primordia. Treatment of J. prismatocarpus seedlings with exogenous auxin or auxin transport inhibitors, which disturb endogenous auxin distribution, eliminated leaf flatness, resulting in a transversally radialized morphology. These treatments did not affect the radialized morphology of leaves of J. wallichianus. Moreover, elimination of leaf flatness by these treatments accompanied dysregulated expression of genetic factors needed to specify the leaf central-marginal polarity in J. prismatocarpus. The findings imply that lamina outgrowth of unifacial leaves relies on proper placement of auxin, which might induce initial leaf flattening and subsequently act to specify leaf polarity, promoting further flattening growth of leaves.
© American Society of Plant Biologists 2021. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures




References
-
- Allen E, Xie Z, Gustafson AM, Carrington JC (2005) microRNA-directed phasing during trans-acting siRNA biogenesis in plants. Cell 121: 207–221 - PubMed
-
- Aloni R, Schwalm K, Langhans M, Ullrich CI (2003) Gradual shifts in sites of free-auxin production during leaf-primordium development and their role in vascular differentiation and leaf morphogenesis in Arabidopsis. Planta 216: 841–853 - PubMed
-
- Bowman JL, Floyd SK (2008) Patterning and polarity in seed plant shoots. Annu Rev Plant Biol 59: 67–88 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

