Turning the spotlight on the oligosaccharide chain of GM1 ganglioside
- PMID: 33620588
- PMCID: PMC7917043
- DOI: 10.1007/s10719-021-09974-y
Turning the spotlight on the oligosaccharide chain of GM1 ganglioside
Abstract
It is well over a century that glycosphingolipids are matter of interest in different fields of research. The hydrophilic oligosaccharide and the lipid moiety, the ceramide, both or separately have been considered in different moments as the crucial portion of the molecule, responsible for the role played by the glycosphingolipids associated to the plasma-membranes or to any other subcellular fraction. Glycosphingolipids are a family of compounds characterized by thousands of structures differing in both the oligosaccharide and the ceramide moieties, but among them, the nervous system monosialylated glycosphingolipid GM1, belonging to the group of gangliosides, has gained particular attention by a multitude of Scientists. In recent years, a series of studies have been conducted on the functional roles played by the hydrophilic part of GM1, its oligosaccharide, that we have named "OligoGM1". These studies allowed to shed new light on the mechanisms underlying the properties of GM1 defining the role of the OligoGM1 in determining precise interactions with membrane proteins instrumental for the neuronal functions, leaving to the ceramide the role of correctly positioning the GM1 in the membrane crucial for the oligosaccharide-protein interactions. In this review we aim to report the recent studies on the cascade of events modulated by OligoGM1, as the bioactive portion of GM1, to support neuronal differentiation and trophism together with preclinical studies on its potential to modify the progression of Parkinson's disease.
Keywords: Drug development; GM1 oligosaccharide; Neurodegeneration; Neurodifferentiation; Neuroprotection; Parkinson’s disease.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
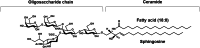






Similar articles
-
Role of the GM1 ganglioside oligosaccharide portion in the TrkA-dependent neurite sprouting in neuroblastoma cells.J Neurochem. 2017 Dec;143(6):645-659. doi: 10.1111/jnc.14146. Epub 2017 Sep 13. J Neurochem. 2017. PMID: 28796418
-
GM1 Ganglioside Is A Key Factor in Maintaining the Mammalian Neuronal Functions Avoiding Neurodegeneration.Int J Mol Sci. 2020 Jan 29;21(3):868. doi: 10.3390/ijms21030868. Int J Mol Sci. 2020. PMID: 32013258 Free PMC article. Review.
-
Gangliosides in the differentiation process of primary neurons: the specific role of GM1-oligosaccharide.Glycoconj J. 2020 Jun;37(3):329-343. doi: 10.1007/s10719-020-09919-x. Epub 2020 Mar 20. Glycoconj J. 2020. PMID: 32198666
-
GM1 Oligosaccharide Crosses the Human Blood-Brain Barrier In Vitro by a Paracellular Route.Int J Mol Sci. 2020 Apr 19;21(8):2858. doi: 10.3390/ijms21082858. Int J Mol Sci. 2020. PMID: 32325905 Free PMC article.
-
GM1 Ganglioside: Past Studies and Future Potential.Mol Neurobiol. 2016 Apr;53(3):1824-1842. doi: 10.1007/s12035-015-9136-z. Epub 2015 Mar 12. Mol Neurobiol. 2016. PMID: 25762012 Review.
Cited by
-
Gangliosides and Cell Surface Ganglioside Metabolic Enzymes in the Nervous System.Adv Neurobiol. 2023;29:305-332. doi: 10.1007/978-3-031-12390-0_11. Adv Neurobiol. 2023. PMID: 36255680
-
A critical role for GM1 ganglioside in the pathophysiology and potential treatment of Parkinson's disease.Glycoconj J. 2022 Feb;39(1):13-26. doi: 10.1007/s10719-021-10002-2. Epub 2021 May 26. Glycoconj J. 2022. PMID: 34037912 Review.
-
Plasma membrane glycosphingolipid signaling: a turning point.Glycoconj J. 2022 Feb;39(1):99-105. doi: 10.1007/s10719-021-10008-w. Epub 2021 Aug 16. Glycoconj J. 2022. PMID: 34398373 Free PMC article.
-
Glycoconjugate journal special issue on: the glycobiology of Parkinson's disease.Glycoconj J. 2022 Feb;39(1):55-74. doi: 10.1007/s10719-021-10024-w. Epub 2021 Nov 10. Glycoconj J. 2022. PMID: 34757539 Review.
-
GM1 ganglioside exerts protective effects against glutamate-excitotoxicity via its oligosaccharide in wild-type and amyotrophic lateral sclerosis motor neurons.FEBS Open Bio. 2023 Dec;13(12):2324-2341. doi: 10.1002/2211-5463.13727. Epub 2023 Nov 15. FEBS Open Bio. 2023. PMID: 37885330 Free PMC article.
References
-
- Chester MA, IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN). Nomenclature of glycolipids–recommendations 1997. Eur. J. Biochem. 1998;257:293–298. - PubMed
-
- Svennerholm L. The Gangliosides. J. Lipid Res. 1964;5:145–55. - PubMed
-
- Schnaar RL. The biology of gangliosides. Adv. Carbohydr. Chem. Biochem. 2019;76:113–148. - PubMed
-
- Wiegandt H. The structure and the function of gangliosides. Angew. Chem. Int. Ed. Engl. 1968;7:87–96. - PubMed
-
- Aureli M, Bassi R, Prinetti A, Chiricozzi E, Pappalardi B, Chigorno V, Di Muzio N, Loberto N, Sonnino S. Ionizing radiations increase the activity of the cell surface glycohydrolases and the plasma membrane ceramide content. Glycoconj. J. 2012;29:585–97. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

