Effects of Spinal Cord Stimulation in Patients with Chronic Nausea, Vomiting, and Refractory Abdominal Pain
- PMID: 33620598
- PMCID: PMC8885488
- DOI: 10.1007/s10620-021-06896-5
Effects of Spinal Cord Stimulation in Patients with Chronic Nausea, Vomiting, and Refractory Abdominal Pain
Abstract
Background: Patients with chronic nausea and vomiting often also have chronic abdominal pain. Spinal cord stimulation (SCS) may provide pain control, but scarce data are available regarding the effect of SCS on chronic nausea and vomiting.
Aims: We aimed to determine the effect of SCS in patients with chronic nausea, vomiting, and refractory abdominal pain.
Methods: Retrospective chart review of 26 consecutive patients who underwent SCS trial for a primary diagnosis of nausea, vomiting and refractory abdominal pain.
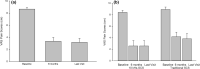
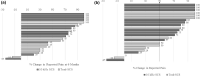
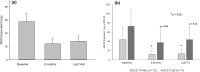
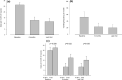
Results: 26 patients underwent SCS trial, with an average age of 48 years. Twenty-three patients (88.5%) reported > 50% pain relief during the temporary SCS trial and then underwent permanent implantation. Patients were then followed for 41 (22-62) months. At baseline, 20 of the 23 patients (87.0%) reported daily nausea, but at 6 months and the most recent follow-up, only 8 (34.8%) and 7 (30.4%) patients, respectively, had daily nausea (p < 0.001). Days of nausea decreased from 26.3 days/month at baseline to 12.8 and 11.7 days/month at 6 months and at the most recent visit, respectively. Vomiting episodes decreased by 50%. Abdominal pain scores improved from 8.7 to 3.0 and 3.2 at 6 months and the most recent visit, respectively (both p < 0.001). Opioid use decreased from 57.7 mg MSO4 equivalents to 24.3 mg at 6 months and to 28.0 mg at the latest patient visit (both p < 0.05).
Conclusions: SCS may be an effective therapy for long-term treatment of symptoms for those patients afflicted with chronic nausea, vomiting, and refractory abdominal pain.
Keywords: Chronic abdominal pain; Chronic nausea and vomiting; Gastric dysmotility; Spinal cord stimulation.
© 2021. The Author(s).
Conflict of interest statement
Dr. Leonardo Kapural reports the following: Research grants from Nevro, Medtronic, Saluda, Biotronik, SPR Therapeutics, Neuros, and Gimer; Consultant for: Nalu, Medtronic, Nevro, Abbott. Dr. Bradbury Brown reports no competing interests. Dr. Shervin Harandi is a Consultant for Nevro. Dr. Jared Rejeski reports no competing interests. Dr. Kenneth Koch reports no competing interests.
Figures
Similar articles
-
Treatment of Chronic Abdominal Pain With 10-kHz Spinal Cord Stimulation: Safety and Efficacy Results From a 12-Month Prospective, Multicenter, Feasibility Study.Clin Transl Gastroenterol. 2020 Feb;11(2):e00133. doi: 10.14309/ctg.0000000000000133. Clin Transl Gastroenterol. 2020. PMID: 32463618 Free PMC article. Clinical Trial.
-
Ten-kHz Spinal Cord Stimulation vs Radiofrequency Ablation of Splanchnic Nerves: A Single-Site Retrospective Comparison of 12-Month Outcomes.Neuromodulation. 2024 Dec;27(8):1449-1456. doi: 10.1016/j.neurom.2024.07.001. Epub 2024 Aug 3. Neuromodulation. 2024. PMID: 39101872
-
Efficacy of gastric electrical stimulation in intractable nausea and vomiting at 10 years: A retrospective analysis of prospectively collected data.Neurogastroenterol Motil. 2020 Nov;32(11):e13949. doi: 10.1111/nmo.13949. Epub 2020 Aug 5. Neurogastroenterol Motil. 2020. PMID: 33107679
-
New advances in the diagnosis and management of nausea and vomiting.Case Manager. 2002 Mar-Apr;13(2):83-6; quiz 87. doi: 10.1067/tcmg.2002.123066. Case Manager. 2002. PMID: 11901376 Review.
-
Treatment-Limiting Complications of Percutaneous Spinal Cord Stimulator Implants: A Review of Eight Years of Experience From an Academic Center Database.Neuromodulation. 2015 Oct;18(7):603-8; discussion 608-9. doi: 10.1111/ner.12312. Epub 2015 Jun 5. Neuromodulation. 2015. PMID: 26053499 Review.
Cited by
-
Spinal Cord Stimulation for Intractable Visceral Pain Originating from the Pelvic and Abdominal Region: A Narrative Review on a Possible New Indication for Patients with Therapy-Resistant Pain.J Pain Res. 2024 Feb 19;17:691-736. doi: 10.2147/JPR.S445616. eCollection 2024. J Pain Res. 2024. PMID: 38405684 Free PMC article. Review.
-
Spinal cord stimulation-induced gastroparesis: A case report.Surg Neurol Int. 2023 Jul 21;14:250. doi: 10.25259/SNI_1133_2022. eCollection 2023. Surg Neurol Int. 2023. PMID: 37560564 Free PMC article.
-
16. Pain in chronic pancreatitis.Pain Pract. 2025 Apr;25(4):e70030. doi: 10.1111/papr.70030. Pain Pract. 2025. PMID: 40189721 Free PMC article. Review.
-
Noninvasive electrical neuromodulation for gastrointestinal motility disorders.Expert Rev Gastroenterol Hepatol. 2023 Dec;17(12):1221-1232. doi: 10.1080/17474124.2023.2288156. Epub 2024 Jan 17. Expert Rev Gastroenterol Hepatol. 2023. PMID: 38018087 Free PMC article. Review.
-
Neuromodulation for functional upper gastrointestinal diseases.Gut Microbiota Integr Wellness. 2023;1:10.54844/gmiw.2022.0087. doi: 10.54844/gmiw.2022.0087. Epub 2023 Apr 28. Gut Microbiota Integr Wellness. 2023. PMID: 38406297 Free PMC article.
References
-
- Hasler WL, Wilson LA, Parkman HP, Koch KL, Abell TL, Nguyen L, Pasricha PJ, Snape WJ, McCallum RW, Sarosiek I, Farrugia G, Calles J, Lee L, Tonascia J, Unalp-Arida A, Hamilton F. Factors related to abdominal pain in gastroparesis: contrast to patients with predominant nausea and vomiting. Neurogastroenterol Motil. 2013;25:427–438. doi: 10.1111/nmo.12091. - DOI - PMC - PubMed
-
- Parkman HP, Yates K, Hasler WL, Nguyen L, Pasricha PJ, Snape WJ, Farrugia G, Koch KL, Calles J, Abell TL, McCallum RW, Lee L, Unalp-Arida A, Tonascia J, Hamilton F, National Institute of Diabetes and Digestive and Kidney Diseases Gastroparesis Clinical Research Consortium Similarities and differences between diabetic and idiopathic gastroparesis. Clin Gastroenterol Hepatol. 2011;9:1056–1064. doi: 10.1016/j.cgh.2011.08.013. - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

