Quantifying cell death induced by doxorubicin, hyperthermia or HIFU ablation with flow cytometry
- PMID: 33623089
- PMCID: PMC7902827
- DOI: 10.1038/s41598-021-83845-2
Quantifying cell death induced by doxorubicin, hyperthermia or HIFU ablation with flow cytometry
Abstract
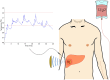
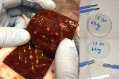
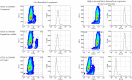
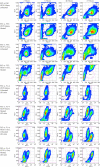
Triggered release and targeted drug delivery of potent anti-cancer agents using hyperthermia-mediated focused-ultrasound (FUS) is gaining momentum in the clinical setting. In early phase studies, tissue biopsy samples may be harvested to assess drug delivery efficacy and demonstrate lack of instantaneous cell death due to FUS exposure. We present an optimised tissue cell recovery method and a cell viability assay, compatible with intra-cellular doxorubicin. Flow cytometry was used to determine levels of cell death with suspensions comprised of: (i) HT29 cell line exposed to hyperthermia (30 min at 47 °C) and/or doxorubicin, or ex-vivo bovine liver tissue exposed to (ii) hyperthermia (up to 2 h at 45 °C), or (iii) ablative high intensity FUS (HIFU). Flow cytometric analysis revealed maximal cell death in HT29 receiving both heat and doxorubicin insults and increases in both cell granularity (p < 0.01) and cell death (p < 0.01) in cells recovered from ex-vivo liver tissue exposed to hyperthermia and high pressures of HIFU (8.2 MPa peak-to-peak free-field at 1 MHz) relative to controls. Ex-vivo results were validated with microscopy using pan-cytokeratin stain. This rapid, sensitive and highly quantitative cell-viability method is applicable to the small masses of liver tissue typically recovered from a standard core biopsy (5-20 mg) and may be applied to tissues of other histological origins including immunostaining.
Conflict of interest statement
The authors declare no competing interests.
Figures
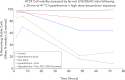
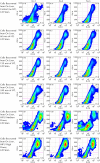
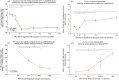


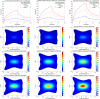
References
-
- Engelhardt R. Rational for clinical application of hyperthermia and drugs. Rev. Med. Chir. Soc. Med. Nat. Iasi. 1987;91(2):347. - PubMed
-
- Kowal CD, Bertino JR. Possible benefits of hyperthermia to chemotherapy. Cancer Res. 1979;39(6):2285–2289. - PubMed
-
- Needham D, Anyarambhatla G, Kong G, Dewhirst MW. A new temperature-sensitive liposome for use with mild hyperthermia: Characterization and testing in a human tumor xenograft model. Cancer Res. 2000;60(5):1197–1201. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

