Intra-Arterial Tissue Plasminogen Activator for Central Retinal Artery Occlusion
- PMID: 33623361
- PMCID: PMC7896758
- DOI: 10.2147/OPTH.S272126
Intra-Arterial Tissue Plasminogen Activator for Central Retinal Artery Occlusion
Abstract
Purpose: To investigate the benefit of early intra-arterial tissue plasminogen activator (IAT) for treatment of central retinal artery occlusion (CRAO).
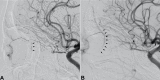
Patients and methods: Fifteen eyes of 15 patients presenting with acute CRAO were included in this retrospective consecutive interventional case series. Patients were excluded if treatment with IAT was not initiated within 12 hours. The diagnosis was confirmed by an ophthalmologist. IAT was performed via a transfemoral arterial approach. Tissue plasminogen activator (tPA) was infused into the ophthalmic artery in aliquots up to 3mg to a maximum of 22mg. Paracentesis was done at the ophthalmologist's discretion. The primary outcome measure was visual acuity after three weeks. Adverse events were recorded during treatment and follow-up visits.
Results: After treatment with IAT, there was a statistically significant improvement in visual acuity, with a mean change of -0.76 (SD 0.91; range -2.4 to 0.85) logMAR (p=0.006). Vision improved by 3 or more lines in 53%, and of these, the mean Snellen visual acuity improvement was >6 lines. Notably, 4 patients (27%) improved from CF or worse to 20/80 or better. The mean dose of tPA used was 17mg and the mean time to treatment was 8.83 hours (range: 5.5 to 12 hours). There were no statistically significant differences based on time to treatment, dose of tPA, or use of a paracentesis. No major adverse events were recorded.
Conclusion: IAT was safe and showed significant visual improvement in this small uncontrolled study. Larger studies and efforts to decrease time to treatment should be initiated to optimize outcomes.
Keywords: CRAO; intervention; ophthalmic artery; tPA; treatment.
© 2021 Sobol et al.
Conflict of interest statement
Dr J Mocco is consultant/investor/stockholder for Endostream, Viseon, Imperative Care, RIST, Synchron, Viz.ai, Perflow, CV Aid, Cerebrotech, Echovate, Rebound, Blink TBI, Serenity, Cardinal Consulting, and NTI; research support for Stryker, Microvention, and Penumbra, outside the submitted work. Dr Richard B Rosen reports personal fees, non-financial support for intellectual property from Optovue; personal fees/grants from Boehringer-Ingelheim, Astellas, Regeneron, Genentech-Roche, NanoRetina, CellView, Bayer, and Teva; non-financial support from OD-OS and Diopsys, personal financial interest from Guardian Health and Opticology, outside the submitted work. The authors report no other conflicts of interest in this work.
Figures



References
LinkOut - more resources
Full Text Sources
Other Literature Sources

