Rpgrip1l controls ciliary gating by ensuring the proper amount of Cep290 at the vertebrate transition zone
- PMID: 33625872
- PMCID: PMC8108517
- DOI: 10.1091/mbc.E20-03-0190
Rpgrip1l controls ciliary gating by ensuring the proper amount of Cep290 at the vertebrate transition zone
Abstract
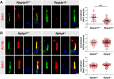
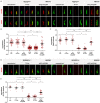
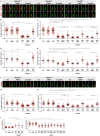
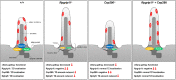
A range of severe human diseases called ciliopathies is caused by the dysfunction of primary cilia. Primary cilia are cytoplasmic protrusions consisting of the basal body (BB), the axoneme, and the transition zone (TZ). The BB is a modified mother centriole from which the axoneme, the microtubule-based ciliary scaffold, is formed. At the proximal end of the axoneme, the TZ functions as the ciliary gate governing ciliary protein entry and exit. Since ciliopathies often develop due to mutations in genes encoding proteins that localize to the TZ, the understanding of the mechanisms underlying TZ function is of eminent importance. Here, we show that the ciliopathy protein Rpgrip1l governs ciliary gating by ensuring the proper amount of Cep290 at the vertebrate TZ. Further, we identified the flavonoid eupatilin as a potential agent to tackle ciliopathies caused by mutations in RPGRIP1L as it rescues ciliary gating in the absence of Rpgrip1l.
Figures

References
-
- Aubusson-Fleury A, Lemullois M, de Loubresse N, Laligné C, Cohen J, Rosnet O, Jerka-Dziadosz M, Beisson J, Koll F (2012). The conserved centrosomal protein FOR20 is required for assembly of the transition zone and basal body docking at the cell surface. J Cell Sci 125, 4395–4404. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

