Sputum sample positivity for Haemophilus influenzae or Moraxella catarrhalis in acute exacerbations of chronic obstructive pulmonary disease: evaluation of association with positivity at earlier stable disease timepoints
- PMID: 33627095
- PMCID: PMC7903661
- DOI: 10.1186/s12931-021-01653-8
Sputum sample positivity for Haemophilus influenzae or Moraxella catarrhalis in acute exacerbations of chronic obstructive pulmonary disease: evaluation of association with positivity at earlier stable disease timepoints
Abstract
Background: Infection with Haemophilus influenzae (Hi) or Moraxella catarrhalis (Mcat) is a risk factor for exacerbation in chronic obstructive pulmonary disease (COPD). The ability to predict Hi- or Mcat-associated exacerbations may be useful for interventions developed to reduce exacerbation frequency.
Methods: In a COPD observational study, sputum samples were collected at monthly stable-state visits and at exacerbation during two years of follow-up. Bacterial species (Hi, Mcat) were identified by culture and quantitative PCR assay. Post-hoc analyses were conducted to assess: (1) first Hi- or Mcat-positive exacerbations given presence or absence of Hi or Mcat at the screening visit (stable-state timepoint); (2) first Hi- or Mcat-positive exacerbations given presence or absence of Hi or Mcat at stable timepoints within previous 90 days; (3) second Hi- or Mcat-positive exacerbations given presence or absence of Hi or Mcat at stable timepoints within previous 90 days. Percentages and risk ratios (RRs) with 95% confidence intervals were calculated.
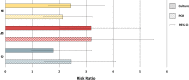
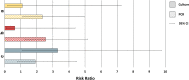
Results: PCR results for analyses 1, 2 and 3 (samples from 84, 88 and 83 subjects, respectively) showed that the risk of an Hi- or Mcat-positive exacerbation is significantly higher if sputum sample was Hi- or Mcat-positive than if Hi- or Mcat-negative at previous stable timepoints (apart from Mcat in analysis 3); RRs ranged from 2.1 to 3.2 for Hi and 1.9 to 2.6 for Mcat.For all analyses, the percentage of Hi- or Mcat-positive exacerbations given previous Hi- or Mcat-positive stable timepoints was higher than the percentage of Hi- or Mcat-positive exacerbations if Hi- or Mcat-negative at previous stable timepoints. Percentage of Hi- or Mcat-positive exacerbations given previous Hi- or Mcat-negative stable timepoints was 26.3%-37.0% for Hi and 17.6%-19.7% for Mcat.
Conclusions: Presence of Hi or Mcat at a stable timepoint was associated with a higher risk of a subsequent Hi- or Mcat-associated exacerbation compared with earlier absence. However, a large percentage of Hi- or Mcat-associated exacerbations was not associated with Hi/Mcat detection at an earlier timepoint. This suggests that administration of an intervention to reduce these exacerbations should be independent of bacterial presence at baseline. Trial Registration https://clinicaltrials.gov/ ; NCT01360398, registered May 25, 2011.
Keywords: Bacterial identification; COPD; Culture; Exacerbation; Haemophilus influenzae; Moraxella catarrhalis; PCR; Vaccination.
Conflict of interest statement
LM, LT, and AKA are employees of the GSK group of companies. AY is a consultant of ICON for the GSK group of companies. LT and AKA hold shares in the GSK group of companies and AKA has a patent PCT/EP2018/071860 – Methods of boosting immune responses – pending to the GSK group of companies. TMAW has received grants from the GSK group of companies during the conduct of this study. TMAW has also received grants from AstraZeneca, Synairgen, and MyMHealth and fees, reimbursement for travel and meeting attendance from Boehringer Ingelheim, Chiesi, and AstraZeneca, outside of the submitted work. In addition, TMAW has a patent 2018 US Patent 62/479562 – Immunogenic Composition, Use and Methods of Treatment – A novel vaccine to prevent exacerbations of COPD pending to the GSK group of companies. TMAW is the Founder and Director of MyMHealth Ltd. All authors declare having no other non-financial relationships and activities.
Figures


Similar articles
-
Ex-vivo RNA expression analysis of vaccine candidate genes in COPD sputum samples.Respir Res. 2023 Oct 5;24(1):243. doi: 10.1186/s12931-023-02525-z. Respir Res. 2023. PMID: 37798723 Free PMC article.
-
Non-typeable Haemophilus influenzae-Moraxella catarrhalis vaccine for the prevention of exacerbations in chronic obstructive pulmonary disease: a multicentre, randomised, placebo-controlled, observer-blinded, proof-of-concept, phase 2b trial.Lancet Respir Med. 2022 May;10(5):435-446. doi: 10.1016/S2213-2600(21)00502-6. Epub 2022 Jan 10. Lancet Respir Med. 2022. PMID: 35026180 Clinical Trial.
-
Impact of bacterial strain acquisition in the lung of patients with COPD: the AERIS study.Infect Dis (Lond). 2022 Nov;54(11):784-793. doi: 10.1080/23744235.2022.2092648. Epub 2022 Jul 6. Infect Dis (Lond). 2022. PMID: 35794793
-
The safety and efficacy of non-typeable Haemophilus influenzae and Moraxella catarrhalis vaccine in chronic obstructive pulmonary disease: a systematic review and meta-analysis of randomized controlled trials.Front Med (Lausanne). 2025 Apr 4;12:1572726. doi: 10.3389/fmed.2025.1572726. eCollection 2025. Front Med (Lausanne). 2025. PMID: 40255595 Free PMC article.
-
[The role of macrolides in treatment of exacerbations of chronic obstructive pulmonary disease].Pol Merkur Lekarski. 2010 Apr;28(166):311-4. Pol Merkur Lekarski. 2010. PMID: 20491344 Review. Polish.
Cited by
-
Ex-vivo RNA expression analysis of vaccine candidate genes in COPD sputum samples.Respir Res. 2023 Oct 5;24(1):243. doi: 10.1186/s12931-023-02525-z. Respir Res. 2023. PMID: 37798723 Free PMC article.
-
Safety and immunogenicity of three doses of non-typeable Haemophilus influenzae-Moraxella catarrhalis (NTHi-Mcat) vaccine when administered according to two different schedules: a phase 2, randomised, observer-blind study.Respir Res. 2022 May 4;23(1):114. doi: 10.1186/s12931-022-02019-4. Respir Res. 2022. PMID: 35509077 Free PMC article. Clinical Trial.
-
Exploring the Role of Gut-Lung Interactions in COPD Pathogenesis: A Comprehensive Review on Microbiota Characteristics and Inflammation Modulation.Chronic Obstr Pulm Dis. 2024 May 29;11(3):311-325. doi: 10.15326/jcopdf.2023.0442. Chronic Obstr Pulm Dis. 2024. PMID: 38563747 Free PMC article.
-
Real-time PCR has advantages over culture-based methods in identifying major airway bacterial pathogens in chronic obstructive pulmonary disease: Results from three clinical studies in Europe and North America.Front Microbiol. 2023 Feb 24;13:1098133. doi: 10.3389/fmicb.2022.1098133. eCollection 2022. Front Microbiol. 2023. PMID: 36909845 Free PMC article.
-
Cytokine Biomarkers of Exacerbations in Sputum From Patients With Chronic Obstructive Pulmonary Disease: A Prospective Cohort Study.J Infect Dis. 2024 Nov 15;230(5):e1112-e1120. doi: 10.1093/infdis/jiae232. J Infect Dis. 2024. PMID: 38836471 Free PMC article.
References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

