Reduction of Neuroinflammation by δ-Opioids Via STAT3-Dependent Pathway in Chronic Glaucoma Model
- PMID: 33628191
- PMCID: PMC7898062
- DOI: 10.3389/fphar.2021.601404
Reduction of Neuroinflammation by δ-Opioids Via STAT3-Dependent Pathway in Chronic Glaucoma Model
Abstract
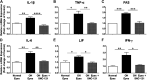
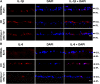
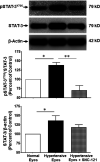
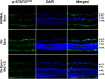
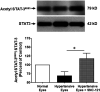
The main objective of this study was to determine the inhibition of pro-inflammatory cytokines and their associated signaling molecules by δ-opioid receptor activation by a selective ligand, SNC-121 in chronic rat glaucoma model. Intraocular pressure was raised in rat eyes by injecting 2 M hypertonic saline into the limbal veins. SNC-121 (1 mg/kg; i. p) or Stattic (5 mg/kg; i. p) was administered in Brown Norway rats daily for 7 days. The mRNA expression of IL-1β, TNF-α, Fas, IL-6, leukemia inhibitory factor, and IFN-γ was increased significantly in the retina of ocular hypertensive animals at day 7, post injury. Administration of SNC-121 (1 mg/kg; i. p. injection) for 7 days (once a day) completely inhibited the increase in the mRNA and protein expression of pro-inflammatory cytokines. Mechanistically, we provide data showing a significant increase in the phosphorylation of STAT3 at tyrosine 705 whereas a moderate but significant increase in the total STAT3 protein expression was also seen in the retina of ocular hypertensive animals. Data illustrated that SNC-121 administration completely abrogated ocular hypertension-induced increase in STAT3Y705 phosphorylation. Interestingly, acetylation of STAT3 at lysine 685 (AcK685) was reduced in ocular hypertensive animals and subsequently increased significantly by SNC-121 treatment. Stattic, a selective STAT3 inhibitor, administration resulted in a complete attenuation in the production of IL-1β and IL-6 in ocular hypertensive animals. In conclusion, δ-opioid receptor activation suppressed the phosphorylation of STAT3 at tyrosine 705 and increased acetylation at lysine 686 and these posttranslational modifications can regulate the production of some but not all pro-inflammatory cytokines in response to glaucomatous injury.
Keywords: glaucoma; neuroinflammantion; opioids; protein acetylation; transcription factors.
Copyright © 2021 Husain, Zaidi, Singh, Guzman and Mehrotra.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Delta-opioid agonist SNC-121 protects retinal ganglion cell function in a chronic ocular hypertensive rat model.Invest Ophthalmol Vis Sci. 2013 Mar 11;54(3):1816-28. doi: 10.1167/iovs.12-10741. Invest Ophthalmol Vis Sci. 2013. PMID: 23404122 Free PMC article.
-
PI3K/Akt Pathway: A Role in δ-Opioid Receptor-Mediated RGC Neuroprotection.Invest Ophthalmol Vis Sci. 2017 Dec 1;58(14):6489-6499. doi: 10.1167/iovs.16-20673. Invest Ophthalmol Vis Sci. 2017. PMID: 29288267 Free PMC article.
-
Changes in Class I and IIb HDACs by δ-Opioid in Chronic Rat Glaucoma Model.Invest Ophthalmol Vis Sci. 2020 Dec 1;61(14):4. doi: 10.1167/iovs.61.14.4. Invest Ophthalmol Vis Sci. 2020. PMID: 33263714 Free PMC article.
-
Histone Deacetylases Regulation by δ-Opioids in Human Optic Nerve Head Astrocytes.Invest Ophthalmol Vis Sci. 2020 Sep 1;61(11):17. doi: 10.1167/iovs.61.11.17. Invest Ophthalmol Vis Sci. 2020. PMID: 32915982 Free PMC article.
-
Toll-Like Receptor 4 (TLR4)/Opioid Receptor Pathway Crosstalk and Impact on Opioid Analgesia, Immune Function, and Gastrointestinal Motility.Front Immunol. 2020 Jul 8;11:1455. doi: 10.3389/fimmu.2020.01455. eCollection 2020. Front Immunol. 2020. PMID: 32733481 Free PMC article. Review.
Cited by
-
The role of the adaptive immune system and T cell dysfunction in neurodegenerative diseases.J Neuroinflammation. 2022 Oct 8;19(1):251. doi: 10.1186/s12974-022-02605-9. J Neuroinflammation. 2022. PMID: 36209107 Free PMC article. Review.
-
Calbindin 2-specific deletion of arginase 2 preserves visual function after optic nerve crush.Cell Death Dis. 2023 Oct 10;14(10):661. doi: 10.1038/s41419-023-06180-6. Cell Death Dis. 2023. PMID: 37816735 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

