Phase 2 Randomized Clinical Study of Netarsudil Ophthalmic Solution in Japanese Patients with Primary Open-Angle Glaucoma or Ocular Hypertension
- PMID: 33629227
- PMCID: PMC8004483
- DOI: 10.1007/s12325-021-01634-9
Phase 2 Randomized Clinical Study of Netarsudil Ophthalmic Solution in Japanese Patients with Primary Open-Angle Glaucoma or Ocular Hypertension
Abstract
Introduction: Netarsudil reduces intraocular pressure (IOP) by increasing aqueous outflow through the trabecular meshwork (TM) pathway and decreasing episcleral venous pressure. The primary objective of this phase 2 study was to evaluate ocular hypotensive efficacy and safety of three netarsudil concentrations (0.01%, 0.02%, and 0.04%) relative to its placebo over 4 weeks in Japanese patients with primary open-angle glaucoma (POAG) or ocular hypertension (OHT).
Methods: Patients were randomized to one of four treatment arms, netarsudil ophthalmic solution 0.01%, 0.02%, 0.04%, or placebo, and treated once-daily (QD) in the evening (P.M.) for 4 weeks. The primary efficacy variable was mean diurnal IOP (average of diurnal time points at 9 A.M., 11 A.M., and 4 P.M.) at week 4.
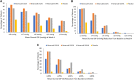
Results: A total of 215 patients were randomized and 207 (96.3%) completed the study. The mean of mean diurnal IOP at baseline ranged from 20.28 to 21.14 mmHg across groups. At week 4, least squares (LS) mean of mean diurnal IOP adjusted for baseline was 16.53, 15.82, 16.06, and 18.94 mmHg in the netarsudil 0.01%, 0.02%, 0.04%, and placebo groups, respectively, demonstrating the superiority of netarsudil (all concentrations) over placebo. At week 4, mean reduction (mean percentage reduction) from baseline in mean diurnal IOP was 4.10 (19.8%), 4.80 (23.5%), 4.81 (23.8%), and 1.73 mmHg (8.2%), respectively, demonstrating statistically significant reductions (p < 0.0001) in all netarsudil concentrations over placebo. Adverse events (AEs) occurred in a concentration-dependent manner, and the incidence of ocular AEs was 34.5%, 42.6%, 68.6%, and 9.1% in the netarsudil 0.01%, 0.02%, 0.04%, and placebo groups, respectively. The most frequently reported AE was conjunctival hyperemia, with an incidence of 23.6%, 37.0%, 56.9%, and 1.8%, respectively. No serious AEs were reported.
Conclusion: Netarsudil ophthalmic solutions 0.01%, 0.02%, and 0.04% dosed QD (P.M.) demonstrated superiority to placebo in terms of hypotensive effectiveness at week 4 and were found to be safe and generally well tolerated. Netarsudil 0.02% QD provided an optimal efficacy and safety profile for the treatment of Japanese patients with POAG or OHT.
Trial registration: NCT03844945.
Keywords: Conjunctival hyperemia; Glaucoma; Intraocular pressure; Netarsudil; Rho-associated protein kinase.
Figures


Similar articles
-
Fixed-Dose Combination of Netarsudil and Latanoprost in Ocular Hypertension and Open-Angle Glaucoma: Pooled Efficacy/Safety Analysis of Phase 3 MERCURY-1 and -2.Adv Ther. 2020 Apr;37(4):1620-1631. doi: 10.1007/s12325-020-01277-2. Epub 2020 Mar 12. Adv Ther. 2020. PMID: 32166538 Free PMC article. Clinical Trial.
-
Phase 3 Clinical Trial Comparing the Safety and Efficacy of Netarsudil to Ripasudil in Patients with Primary Open-Angle Glaucoma or Ocular Hypertension: Japan Rho Kinase Elevated Intraocular Pressure Treatment Trial (J-ROCKET).Adv Ther. 2023 Oct;40(10):4639-4656. doi: 10.1007/s12325-023-02550-w. Epub 2023 Aug 21. Adv Ther. 2023. PMID: 37603205 Free PMC article. Clinical Trial.
-
Netarsudil Improves Trabecular Outflow Facility in Patients with Primary Open Angle Glaucoma or Ocular Hypertension: A Phase 2 Study.Am J Ophthalmol. 2021 Jun;226:262-269. doi: 10.1016/j.ajo.2021.01.019. Epub 2021 Jan 29. Am J Ophthalmol. 2021. PMID: 33524367 Clinical Trial.
-
Netarsudil ophthalmic solution 0.02% for the treatment of patients with open-angle glaucoma or ocular hypertension.Drugs Today (Barc). 2018 Aug;54(8):467-478. doi: 10.1358/dot.2018.54.8.2849627. Drugs Today (Barc). 2018. PMID: 30209441 Review.
-
[ROCK (RHO-KINASE INHIBITORS) FOR THE TREATMENT OF OPEN-ANGLE GLAUCOMA AND OCULAR HYPERTENSION].Harefuah. 2023 Mar;162(3):160-164. Harefuah. 2023. PMID: 36966373 Review. Hebrew.
Cited by
-
Comparative Efficacy and Safety of Netarsudil-Containing Interventions for Intraocular Pressure Control: A Systematic Review and Network Meta-Analysis.Cureus. 2025 Feb 3;17(2):e78432. doi: 10.7759/cureus.78432. eCollection 2025 Feb. Cureus. 2025. PMID: 39902256 Free PMC article. Review.
-
Pediatric Glaucoma-From Screening, Early Detection to Management.Children (Basel). 2023 Jan 18;10(2):181. doi: 10.3390/children10020181. Children (Basel). 2023. PMID: 36832310 Free PMC article. Review.
-
Intraocular pressure-lowering efficacy and ocular safety of Rho-kinase inhibitor in glaucoma: a meta-analysis and systematic review of prospective randomized trials.Graefes Arch Clin Exp Ophthalmol. 2022 Mar;260(3):937-948. doi: 10.1007/s00417-021-05379-7. Epub 2021 Sep 7. Graefes Arch Clin Exp Ophthalmol. 2022. PMID: 34491427
-
Rho Kinase (ROCK) Inhibitors in the Treatment of Glaucoma and Glaucoma Surgery: A Systematic Review of Early to Late Phase Clinical Trials.Pharmaceuticals (Basel). 2025 Apr 3;18(4):523. doi: 10.3390/ph18040523. Pharmaceuticals (Basel). 2025. PMID: 40283958 Free PMC article. Review.
-
Medical and surgical treatment management in open angle glaucoma patients of Asian descent: A narrative review.Eur J Ophthalmol. 2025 Sep;35(5):1883-1895. doi: 10.1177/11206721251340435. Epub 2025 May 19. Eur J Ophthalmol. 2025. PMID: 40388924 Free PMC article. Review.
References
-
- Japan Glaucoma Society Glaucoma Practice Guidelines Development Committee. Glaucoma medical care guideline 4th edition. Nichigankaishi. 2018;122:5–53.
-
- Iwase A, Suzuki Y, Araie M, et al. The prevalence of primary open-angle glaucoma in Japanese: the Tajimi study. Ophthalmology. 2004;111:1641–1648. - PubMed
-
- Yamamoto T, Iwase A, Araie M, et al. Prevalence of primary angle closure and secondary glaucoma in a Japanese population. Ophthalmology. 2005;112:1661–1669. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

