Recent Advances in Photoredox-Mediated Radical Conjugate Addition Reactions: An Expanding Toolkit for the Giese Reaction
- PMID: 33629454
- PMCID: PMC8382814
- DOI: 10.1002/anie.202016666
Recent Advances in Photoredox-Mediated Radical Conjugate Addition Reactions: An Expanding Toolkit for the Giese Reaction
Abstract
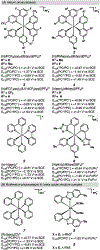
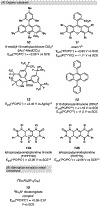
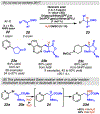
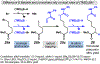
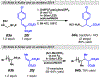
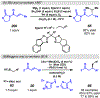
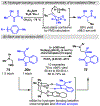
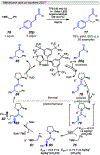
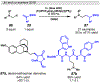
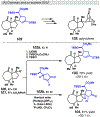
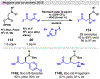
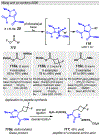
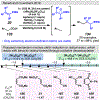
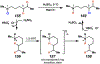
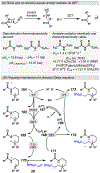
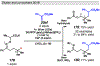
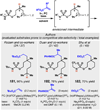
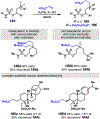
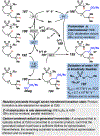
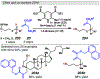
Photomediated Giese reactions are at the forefront of radical chemistry, much like the classical tin-mediated Giese reactions were nearly forty years ago. With the global recognition of organometallic photocatalysts for the mild and tunable generation of carbon-centered radicals, chemists have developed a torrent of strategies to form previously inaccessible radical intermediates that are capable of engaging in intermolecular conjugate addition reactions. This Review summarizes advances in photoredox-mediated Giese reactions since 2013, with a focus on the breadth of methods that provide access to crucial carbon-centered radical intermediates that can engage in radical conjugate addition processes.
Keywords: Giese reaction; photocatalysis; photoredox catalysis; radical Michael addition; radical conjugate addition.
© 2021 Wiley-VCH GmbH.
Figures








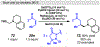




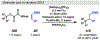
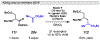





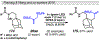




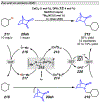
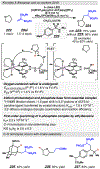
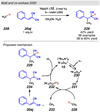
References
-
- For the seminal publications establishing the synthetic utility of radical conjugate addition reactions, see: Giese B, Lachhein S, Angew. Chem. Int. Ed 1981, 20, 967
- Angew. Chem 1981, 93, 1016.
- Giese B, Dupuis J, Angew. Chem. Int. Ed 1983, 22, 622–623
- Angew. Chem 1983, 95, 633–634.
- Giese B, Gonzalez-Gomez JA, Witzel T, Angew. Chem. Int. Ed 1984, 23, 69–70
- Angew. Chem 1984, 96, 51–52.
-
- Studer A, Curran DP, Angew. Chem. Int. Ed 2016, 55, 58–102 - PubMed
- Angew. Chem 2016, 128, 58–106.
-
- For reviews demonstrating the scope and selectivity of the Giese reaction, see: Giese B, Angew. Chem. Int. Ed 1983, 22, 753–764
- Angew. Chem 1983, 95, 771–782.
- Giese B, Angew. Chem. Int. Ed 1985, 24, 553–565
- Angew. Chem 1983, 97, 555–567.
- Sibi MK, Venkatraman L, Liu M, J. Am. Chem. Soc 2001, 123, 8444–8445. - PubMed
- Bar G, Parsons AF, Chem. Soc. Rev 2003, 32, 251–263. - PubMed
- Srikanth GSC, Castle SL, Tetrahedron, 2005, 61, 10377–10441.
-
- For seminal examples of the application of metal polypyridal complexes in synthetic transformations, see: Hedstrand DM, Kruizinga WM, Kellogg RM, Tetrahedron Lett. 1978, 19, 1255–1258.
- Pac C, Ihama M, Yasuda M, Miyauchi Y, Sakurai H, J. Am. Chem. Soc 1981, 103, 6495–6497.
- Hironaka K, Fukuzumi S, Tanaka T, J. Chem. Soc., Perkin Trans. 2 1984, 1705–1709.
- Cano-Yelo H, Deronzier A, J. Chem. Soc. Perkin Trans. II, 1984, 1093–1098.
-
- For select reviews on photoredox-mediated processes published since 2013, see: Staveness D, Bosque I, Stephenson CRJ, Acc. Chem. Res 2016, 49, 2295–2306. - PMC - PubMed
- Uygur M, Mancheño OG, Org. Biomol. Chem 2019, 17, 5475–5489. - PubMed
- Romero NA, Nicewicz DA, Chem. Rev 2016, 116, 10075–10166. - PubMed
- Schultz DM, Yoon TP, Science, 2014, 343, 1239176. - PMC - PubMed
- Michelin C, Hoffmann N, ACS Catal. 2018, 8, 12046–12055
- McAtee RC, McCLain EJ, Stephenson CRJ, Trends in Chemistry, 2019, 1, 111–125. - PMC - PubMed
- Teegardin K, Day JI, Chan J, Weaver J, Or. Process Res. Dev 2016, 20, 1156–1163. - PMC - PubMed
- Cavedon C, Seeberger PH, Pieber B, Eur. J. Org. Chem 2019, 1–15. - PMC - PubMed
- Skubi KL, Blum TR, Yoon TP, Chem. Rev 2016, 116, 10035–10074. - PMC - PubMed
- Fensterbank L, Goddard J-P, Ollivier C in Visible Light Photocatalysis in Organic Chemistry; (Eds.; Stephenson CRJ, Yoon TP, MacMillan DWC)Wiley, Weinheim, Germany, 2018, pp 25–59.
- Akita M, Kioke T. C.R. Chimie, 2015, 18, 742–751.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

