The Role of the Dopamine Transporter in the Effects of Amphetamine on Sleep and Sleep Architecture in Drosophila
- PMID: 33630236
- PMCID: PMC8384956
- DOI: 10.1007/s11064-021-03275-4
The Role of the Dopamine Transporter in the Effects of Amphetamine on Sleep and Sleep Architecture in Drosophila
Abstract
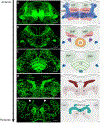
The dopamine transporter (DAT) mediates the inactivation of released dopamine (DA) through its reuptake, and thereby plays an important homeostatic role in dopaminergic neurotransmission. Amphetamines exert their stimulant effects by targeting DAT and inducing the reverse transport of DA, leading to a dramatic increase of extracellular DA. Animal models have proven critical to investigating the molecular and cellular mechanisms underlying transporter function and its modulation by psychostimulants such as amphetamine. Here we establish a behavioral model for amphetamine action using adult Drosophila melanogaster. We use it to characterize the effects of amphetamine on sleep and sleep architecture. Our data show that amphetamine induces hyperactivity and disrupts sleep in a DA-dependent manner. Flies that do not express a functional DAT (dDAT null mutants) have been shown to be hyperactive and to exhibit significantly reduced sleep at baseline. Our data show that, in contrast to its action in control flies, amphetamine decreases the locomotor activity of dDAT null mutants and restores their sleep by modulating distinct aspects of sleep structure. To begin to explore the circuitry involved in the actions of amphetamine on sleep, we also describe the localization of dDAT throughout the fly brain, particularly in neuropils known to regulate sleep. Together, our data establish Drosophila as a robust model for studying the regulatory mechanisms that govern DAT function and psychostimulant action.
Keywords: Adult brain; Behavior; Dopamine transporter localization; Drosophila melanogaster; Genetics; Locomotion; Psychostimulants; Sleep.
© 2021. The Author(s), under exclusive licence to Springer Science+Business Media, LLC part of Springer Nature.
Figures
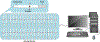

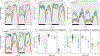
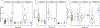

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

