Synthetic Enzyme-Catalyzed CO2 Fixation Reactions
- PMID: 33631048
- PMCID: PMC8252502
- DOI: 10.1002/cssc.202100159
Synthetic Enzyme-Catalyzed CO2 Fixation Reactions
Abstract
In recent years, (de)carboxylases that catalyze reversible (de)carboxylation have been targeted for application as carboxylation catalysts. This has led to the development of proof-of-concept (bio)synthetic CO2 fixation routes for chemical production. However, further progress towards industrial application has been hampered by the thermodynamic constraint that accompanies fixing CO2 to organic molecules. In this Review, biocatalytic carboxylation methods are discussed with emphases on the diverse strategies devised to alleviate the inherent thermodynamic constraints and their application in synthetic CO2 -fixation cascades.
Keywords: CO2 fixation, enzymes; biocatalysis; carboxylation; cascade reactions.
© 2021 The Authors. ChemSusChem published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







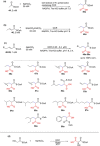

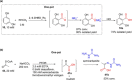

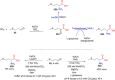
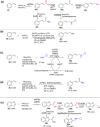
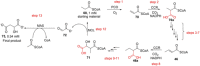
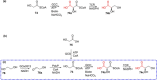


References
-
- Artz J., Müller T. E., Thenert K., Kleinekorte J., Meys R., Sternberg A., Bardow A., Leitner W., Chem. Rev. 2018, 118, 434–504. - PubMed
-
- Olah G. A., Prakash G. K. S., Goeppert A., J. Am. Chem. Soc. 2011, 133, 12881–12898. - PubMed
-
- Liu Y., Li Y., Chen Y., Qu T., Shu C., Yang X., Zhu H., Guo S., Zhao S., Asefa T., Liu Y., J. Mater. Chem. A 2020, 8, 8329–8336.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

