Iron Oxide Nanoparticles Combined with Cytosine Arabinoside Show Anti-Leukemia Stem Cell Effects on Acute Myeloid Leukemia by Regulating Reactive Oxygen Species
- PMID: 33633448
- PMCID: PMC7900778
- DOI: 10.2147/IJN.S278885
Iron Oxide Nanoparticles Combined with Cytosine Arabinoside Show Anti-Leukemia Stem Cell Effects on Acute Myeloid Leukemia by Regulating Reactive Oxygen Species
Abstract
Background and aim: Acute myeloid leukemia (AML), initiated and maintained by leukemia stem cells (LSCs), is often relapsed or refractory to therapy. The present study aimed at assessing the effects of nanozyme-like Fe3O4 nanoparticles (IONPs) combined with cytosine arabinoside (Ara-C) on LSCs in vitro and in vivo.
Methods: The CD34+CD38-LSCs, isolated from human AML cell line KG1a by a magnetic activated cell sorting method, were treated with Ara-C, IONPs, and Ara-C+ IONPs respectively in vitro. The cellular proliferation, apoptosis, reactive oxygen species (ROS), and the related molecular expression levels in LSCs were analyzed using flow cytometry, RT-qPCR, and Western blot. The nonobese diabetic/severe combined immune deficiency mice were transplanted with LSCs or non-LSCs via tail vein, and then the mice were treated with Ara-C, IONPs and IONPs plus Ara-C, respectively. The therapeutic effects on the AML bearing mice were further evaluated.
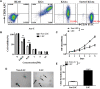
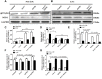
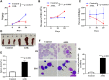
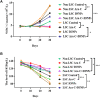
Results: LSCs indicated stronger cellular proliferation, more clone formation, and more robust resistance to Ara-C than non-LSCs. Compared with LSCs treated with Ara-C alone, LSCs treated with IONPs plus Ara-C showed a significant increase in apoptosis and ROS levels that might be regulated by nanozyme-like IONPs via improving the expression of pro-oxidation molecule gp91-phox but decreasing the expression of antioxidation molecule superoxide dismutase 1. The in vivo results suggested that, compared with the AML bearing mice treated with Ara-C alone, the mice treated with IONPs plus Ara-C markedly reduced the abnormal leukocyte numbers in peripheral blood and bone marrow and significantly extended the survival of AML bearing mice.
Conclusion: IONPs combined with Ara-C showed the effectiveness on reducing AML burden in the mice engrafted with LSCs and extending mouse survival by increasing LSC's ROS level to induce LSC apoptosis. Our findings suggest that targeting LSCs could control the AML relapse by using IONPs plus Ara-C.
Keywords: Fe3O4 nanoparticles; acute myeloid leukemia; cytosine arabinoside; leukemia stem cells; reactive oxygen species.
© 2021 Dou et al.
Conflict of interest statement
The authors declare no conflicts of interest in this work.
Figures

Similar articles
-
[Effectiveness of anti-CD47 antibody and Ara-C combination targeting therapy NOD/SCID mouse of myeloid leukemia].Zhongguo Dang Dai Er Ke Za Zhi. 2013 Jul;15(7):577-82. Zhongguo Dang Dai Er Ke Za Zhi. 2013. PMID: 23866283 Chinese.
-
Disulfiram/copper selectively eradicates AML leukemia stem cells in vitro and in vivo by simultaneous induction of ROS-JNK and inhibition of NF-κB and Nrf2.Cell Death Dis. 2017 May 18;8(5):e2797. doi: 10.1038/cddis.2017.176. Cell Death Dis. 2017. PMID: 28518151 Free PMC article.
-
SYK inhibition targets acute myeloid leukemia stem cells by blocking their oxidative metabolism.Cell Death Dis. 2020 Nov 6;11(11):956. doi: 10.1038/s41419-020-03156-8. Cell Death Dis. 2020. PMID: 33159047 Free PMC article.
-
Harnessing Azelaic Acid for Acute Myeloid Leukemia Treatment: A Novel Approach to Overcoming Chemoresistance and Improving Outcomes.Int J Mol Sci. 2025 May 3;26(9):4362. doi: 10.3390/ijms26094362. Int J Mol Sci. 2025. PMID: 40362596 Free PMC article. Review.
-
Extinguishing the Embers: Targeting AML Metabolism.Trends Mol Med. 2021 Apr;27(4):332-344. doi: 10.1016/j.molmed.2020.10.001. Epub 2020 Oct 26. Trends Mol Med. 2021. PMID: 33121874 Free PMC article. Review.
Cited by
-
Chlorambucil-Iron Oxide Nanoparticles as a Drug Delivery System for Leukemia Cancer Cells.Int J Nanomedicine. 2021 Sep 8;16:6205-6216. doi: 10.2147/IJN.S312752. eCollection 2021. Int J Nanomedicine. 2021. PMID: 34526768 Free PMC article.
-
Eltrombopag and its iron chelating properties in pediatric acute myeloid leukemia.Oncotarget. 2021 Jul 6;12(14):1377-1387. doi: 10.18632/oncotarget.28000. eCollection 2021 Jul 6. Oncotarget. 2021. PMID: 34262648 Free PMC article.
-
[Advances of ferroptosis pathways in acute myeloid leukemia].Zhonghua Xue Ye Xue Za Zhi. 2023 Jun 14;44(6):525-528. doi: 10.3760/cma.j.issn.0253-2727.2023.06.019. Zhonghua Xue Ye Xue Za Zhi. 2023. PMID: 37550216 Free PMC article. Chinese. No abstract available.
-
Application of iron oxide nanoparticles in the diagnosis and treatment of leukemia.Front Pharmacol. 2023 Mar 30;14:1177068. doi: 10.3389/fphar.2023.1177068. eCollection 2023. Front Pharmacol. 2023. PMID: 37063276 Free PMC article. Review.
References
-
- Guo J, Russell EG, Darcy R, et al. Antibody-targeted cyclodextrin-based nanoparticles for siRNA delivery in the treatment of acute myeloid leukemia: physicochemical characteristics, in vitro mechanistic studies, and ex vivo patient derived therapeutic efficacy. Mol Pharm. 2017;14(3):940–952. doi:10.1021/acs.molpharmaceut.6b01150 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

