Preventing Bone Stress Injuries in Runners with Optimal Workload
- PMID: 33635519
- PMCID: PMC8316280
- DOI: 10.1007/s11914-021-00666-y
Preventing Bone Stress Injuries in Runners with Optimal Workload
Abstract
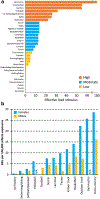
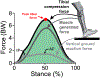
Bone stress injuries (BSIs) occur at inopportune times to invariably interrupt training. All BSIs in runners occur due to an "error" in workload wherein the interaction between the number and magnitude of bone tissue loading cycles exceeds the ability of the tissue to resist the repetitive loads. There is not a single optimal bone workload, rather a range which is influenced by the prevailing scenario. In prepubertal athletes, optimal bone workload consists of low-repetitions of fast, high-magnitude, multidirectional loads introduced a few times per day to induce bone adaptation. Premature sports specialization should be avoided so as to develop a robust skeleton that is structurally optimized to withstand multidirectional loading. In the mature skeleton, optimal workload enables gains in running performance but minimizes bone damage accumulation by sensibly progressing training, particularly training intensity. When indicated (e.g., following repeated BSIs), attempts to reduce bone loading magnitude should be considered, such as increasing running cadence. Determining the optimal bone workload for an individual athlete to prevent and manage BSIs requires consistent monitoring. In the future, it may be possible to clinically determine bone loads at the tissue level to facilitate workload progressions and prescriptions.
Keywords: Exercise; Relative energy deficiency in sport; Running; Stress fracture; Stress reaction.
© 2021. The Author(s), under exclusive licence to Springer Science+Business Media, LLC part of Springer Nature.
Conflict of interest statement
Figures


References
-
- Burr DB, Forwood MR, Fyhrie DP, Martin RB, Schaffler MB, Turner CH. Bone microdamage and skeletal fragility in osteoporotic and stress fractures. J Bone Miner Res. 1997;12:6–15. - PubMed
-
- Burr DB. Targeted and nontargeted remodeling. Bone. 2002;30(1):2–4. - PubMed
-
- Nattiv A, Kennedy G, Barrack MT, Abdelkerim A, Goolsby MA, Arends JC et al. Correlation of MRI grading of bone stress injuries with clinical risk factors and return to play: a 5-year prospective study in collegiate track and field athletes. Am J Sports Med. 2013;41(8):1930–41. doi:10.1177/0363546513490645. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

