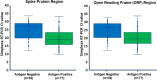
Evaluation of Performance of the BD Veritor SARS-CoV-2 Chromatographic Immunoassay Test in Patients with Symptoms of COVID-19
- PMID: 33637583
- PMCID: PMC8091854
- DOI: 10.1128/JCM.00260-21
Evaluation of Performance of the BD Veritor SARS-CoV-2 Chromatographic Immunoassay Test in Patients with Symptoms of COVID-19
Keywords: COVID-19; Simplexa RT-PCR; antigen test; the Veritor test.
Figures
Comment on
-
Clinical Evaluation of BD Veritor SARS-CoV-2 Point-of-Care Test Performance Compared to PCR-Based Testing and versus the Sofia 2 SARS Antigen Point-of-Care Test.J Clin Microbiol. 2020 Dec 17;59(1):e02338-20. doi: 10.1128/JCM.02338-20. Print 2020 Dec 17. J Clin Microbiol. 2020. PMID: 33023911 Free PMC article.
References
-
- World Health Organization. 2020. WHO announces COVID-19 outbreak a pandemic. WHO Regional Office for Europe, Copenhagen, Denmark. https://www.euro.who.int/en/health-topics/health-emergencies/coronavirus....
-
- Nagura-Ikeda M, Imai K, Tabata S, Miyoshi K, Murahara N, Mizuno T, Horiuchi M, Kato K, Imoto Y, Iwata M, Mimura S, Ito T, Tamura K, Kato Y. 2020. Clinical evaluation of self-collected saliva by quantitative reverse transcription-PCR (RT-qPCR), direct RT-qPCR, reverse transcription-loop-mediated isothermal amplification, and a rapid antigen test to diagnose COVID-19. J Clin Microbiol 58:e01438-20. 10.1128/JCM.01438-20. - DOI - PMC - PubMed
-
- Bordi L, Piralla A, Lalle E, Giardina F, Colavita F, Tallarita M, Sberna G, Novazzi F, Meschi S, Castilletti C, Brisci A, Minnucci G, Tettamanzi V, Baldanti F, Capobianchi MR. 2020. Rapid and sensitive detection of SARS-CoV-2 RNA using the Simplexa COVID-19 direct assay. J Clin Virol 128:104416. 10.1016/j.jcv.2020.104416. - DOI - PMC - PubMed
-
- Centers for Disease Control and Prevention. 2020. Interim guidance for antigen testing for SARS-CoV-2. CDC, Atlanta, GA. https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antigen-tests-gu....
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous