Mixture Cure Models in Oncology: A Tutorial and Practical Guidance
- PMID: 33638063
- PMCID: PMC8160049
- DOI: 10.1007/s41669-021-00260-z
Mixture Cure Models in Oncology: A Tutorial and Practical Guidance
Abstract
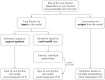
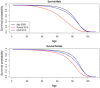
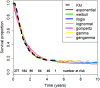
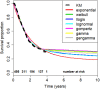
Novel cancer therapies are associated with survival patterns that differ from established therapies, which may include survival curves that plateau after a certain follow-up time point. A fraction of the patient population is then considered statistically cured and subject to the same mortality experience as the cancer-free general population. Mixture cure models have been developed to account for this characteristic. As compared to standard survival analysis, mixture cure models can often lead to profoundly different estimates of long-term survival, required for health economic evaluations. This tutorial is designed as a practical introduction to mixture cure models. Step-by-step instructions are provided for the entire implementation workflow, i.e., from gathering and combining data from different sources to fitting models using maximum likelihood estimation and model results interpretation. Two mixture cure models were developed to illustrate (1) an "uninformed" approach where the cure fraction is estimated from trial data and (2) an "informed" approach where the cure fraction is obtained from an external source (e.g., real-world data) used as an input to the model. These models were implemented in the statistical software R, with the freely available code on GitHub. The cure fraction can be estimated as an output from ("uninformed" approach) or used as an input to ("informed" approach) a mixture cure model. Mixture cure models suggest presumed estimates of long-term survival proportions, especially in instances where some fraction of patients is expected to be statistically cured. While this type of model may initially seem complex, it is straightforward to use and interpret. Mixture cure models have the potential to improve the accuracy of survival estimates for treatments associated with statistical cure, and the present tutorial outlines the interpretation and implementation of mixture cure models in R. This type of model will likely become more widely used in health economic analyses as novel cancer therapies enter the market.
Conflict of interest statement
JR is an employee of F. Hoffmann-La Roche, which develops and markets pharmaceutical products in oncology, including vemurafenib and cobimetinib, which are used as examples in this tutorial. JP was an employee of Ossian Health Economics and Communications, which received consulting fees from F. Hoffmann-La Roche to support the preparation of this tutorial. NP and FF were employees of F. Hoffmann-La Roche. The authors declare that they have no other competing interests.
Figures

References
-
- Collaboration, G.B.o.D.C. et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2016: a systematic analysis for the global burden of disease study. JAMA Oncol. 2018;4(11):1553–1568. doi: 10.1001/jamaoncol.2018.2706. - DOI - PMC - PubMed
-
- Allemani C, et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet (London, England) 2018;391(10125):1023–1075. doi: 10.1016/S0140-6736(17)33326-3. - DOI - PMC - PubMed
-
- Ventola CL. Cancer immunotherapy, part 1: current strategies and agents. P & T : a peer-reviewed journal for formulary management 2017;42(6):375–383. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5440098/ - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

