Fungal G-Protein-Coupled Receptors: A Promising Mediator of the Impact of Extracellular Signals on Biosynthesis of Ochratoxin A
- PMID: 33643259
- PMCID: PMC7907439
- DOI: 10.3389/fmicb.2021.631392
Fungal G-Protein-Coupled Receptors: A Promising Mediator of the Impact of Extracellular Signals on Biosynthesis of Ochratoxin A
Abstract
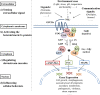
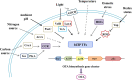
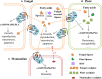
G-protein-coupled receptors (GPCRs) are transmembrane receptors involved in transducing signals from the external environment inside the cell, which enables fungi to coordinate cell transport, metabolism, and growth to promote their survival, reproduction, and virulence. There are 14 classes of GPCRs in fungi involved in sensing various ligands. In this paper, the synthesis of mycotoxins that are GPCR-mediated is discussed with respect to ligands, environmental stimuli, and intra-/interspecific communication. Despite their apparent importance in fungal biology, very little is known about the role of ochratoxin A (OTA) biosynthesis by Aspergillus ochraceus and the ligands that are involved. Fortunately, increasing evidence shows that the GPCR that involves the AF/ST (sterigmatocystin) pathway in fungi belongs to the same genus. Therefore, we speculate that GPCRs play an important role in a variety of environmental signals and downstream pathways in OTA biosynthesis. The verification of this inference will result in a more controllable GPCR target for control of fungal contamination in the future.
Keywords: G protein-coupled receptors; ochratoxin A; oxylipin; quorum sensing; trans-kingdom; transcription factor.
Copyright © 2021 Gao, Xu, Huang and Liang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

