Intestinal and systemic inflammation induced by symptomatic and asymptomatic enterotoxigenic E. coli infection and impact on intestinal colonization and ETEC specific immune responses in an experimental human challenge model
- PMID: 33645430
- PMCID: PMC7919917
- DOI: 10.1080/19490976.2021.1891852
Intestinal and systemic inflammation induced by symptomatic and asymptomatic enterotoxigenic E. coli infection and impact on intestinal colonization and ETEC specific immune responses in an experimental human challenge model
Abstract
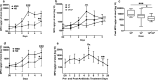
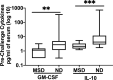
Recent studies have gained a better appreciation of the potential impacts of enteric infections beyond symptomatic diarrhea. It is recognized that infections by several enteropathogens could be associated with growth deficits in children and intestinal and systemic inflammation may play an important underlying role. With enterotoxigenic E. coli (ETEC) being one of the leading causes of diarrhea among children in the developing world and important contributor to stunting, a better understanding of the impact of ETEC infection beyond diarrhea is timely and greatly needed. To address this, we evaluated if ETEC infection induces intestinal and systemic inflammation and its impact on colonization and immune responses to ETEC vaccine-specific antigens in a dose descending experimental human challenge model using ETEC strain H10407. This study demonstrates that the concentrations of myeloperoxidase (MPO) in stool and intestinal fatty acid-binding protein (an indicator of compromised intestinal epithelial integrity) in serum, significantly increased following ETEC infection in both diarrhea and asymptomatic cases and the magnitudes and kinetics of MPO are dose and clinical outcome dependent. Cytokines IL-17A and IFN-γ were significantly increased in serum post-ETEC challenge. In addition, higher pre-challenge concentrations of cytokines IL-10 and GM-CSF were associated with protection from ETEC diarrhea. Interestingly, higher MPO concentrations were associated with higher intestinal colonization of ETEC and lower seroconversions of colonization factor I antigen, but the reverse was noted for seroconversions to heat-labile toxin B-subunit. Together this study has important implications for understanding the acute and long-term negative health outcomes associated with ETEC infection.
Keywords: ETEC; cytokines; immune response; intestinal fatty acid-binding protein; intestinal inflammation; myeloperoxidase; systemic inflammation.
Figures




Similar articles
-
Attenuated Escherichia coli strains expressing the colonization factor antigen I (CFA/I) and a detoxified heat-labile enterotoxin (LThK63) enhance clearance of ETEC from the lungs of mice and protect mice from intestinal ETEC colonization and LT-induced fluid accumulation.Vet Immunol Immunopathol. 2013 Mar 15;152(1-2):57-67. doi: 10.1016/j.vetimm.2012.10.001. Epub 2012 Oct 5. Vet Immunol Immunopathol. 2013. PMID: 23122616
-
Preexisting antibodies to homologous colonization factors and heat-labile toxin in serum, and the risk to develop enterotoxigenic Escherichia coli-associated diarrhea.Diagn Microbiol Infect Dis. 2008 Feb;60(2):229-31. doi: 10.1016/j.diagmicrobio.2007.09.010. Epub 2007 Oct 25. Diagn Microbiol Infect Dis. 2008. PMID: 17964106
-
Characterization of Mucosal Immune Responses to Enterotoxigenic Escherichia coli Vaccine Antigens in a Human Challenge Model: Response Profiles after Primary Infection and Homologous Rechallenge with Strain H10407.Clin Vaccine Immunol. 2015 Nov 18;23(1):55-64. doi: 10.1128/CVI.00617-15. Print 2016 Jan. Clin Vaccine Immunol. 2015. PMID: 26581889 Free PMC article. Clinical Trial.
-
Review on pathogenicity mechanism of enterotoxigenic Escherichia coli and vaccines against it.Microb Pathog. 2018 Apr;117:162-169. doi: 10.1016/j.micpath.2018.02.032. Epub 2018 Feb 21. Microb Pathog. 2018. PMID: 29474827 Review.
-
Current Progress in Developing Subunit Vaccines against Enterotoxigenic Escherichia coli-Associated Diarrhea.Clin Vaccine Immunol. 2015 Sep;22(9):983-91. doi: 10.1128/CVI.00224-15. Epub 2015 Jul 1. Clin Vaccine Immunol. 2015. PMID: 26135975 Free PMC article. Review.
Cited by
-
Sargramostim for Prophylactic Management of Gastrointestinal Immune-Related Adverse Events of Immune Checkpoint Inhibitor Therapy for Cancer.Cancers (Basel). 2024 Jan 24;16(3):501. doi: 10.3390/cancers16030501. Cancers (Basel). 2024. PMID: 38339253 Free PMC article. Review.
-
Dietary Bacteriophage Administration Alleviates Enterotoxigenic Escherichia coli-Induced Diarrhea and Intestinal Impairment through Regulating Intestinal Inflammation and Gut Microbiota in a Newly Weaned Mouse Model.Int J Mol Sci. 2024 Oct 5;25(19):10736. doi: 10.3390/ijms251910736. Int J Mol Sci. 2024. PMID: 39409065 Free PMC article.
-
Protocatechuic acid and quercetin attenuate ETEC-caused IPEC-1 cell inflammation and injury associated with inhibition of necroptosis and pyroptosis signaling pathways.J Anim Sci Biotechnol. 2023 Feb 1;14(1):5. doi: 10.1186/s40104-022-00816-x. J Anim Sci Biotechnol. 2023. PMID: 36721159 Free PMC article.
-
Efficacy of an Enterotoxigenic Escherichia coli (ETEC) Vaccine on the Incidence and Severity of Traveler's Diarrhea (TD): Evaluation of Alternative Endpoints and a TD Severity Score.Microorganisms. 2023 Sep 27;11(10):2414. doi: 10.3390/microorganisms11102414. Microorganisms. 2023. PMID: 37894071 Free PMC article.
-
Inflammation, the kynurenines, and mucosal injury during human experimental enterotoxigenic Escherichia coli infection.Med Microbiol Immunol. 2024 Mar 2;213(1):2. doi: 10.1007/s00430-024-00786-z. Med Microbiol Immunol. 2024. PMID: 38430452 Free PMC article.
References
-
- Chakraborty S, Harro C, DeNearing B, Brubaker J, Connor S, Maier N, Dally L, Flores J, Bourgeois AL, Walker R, et al. Impact of lower challenge doses of enterotoxigenic Escherichia coli on clinical outcome, intestinal colonization and immune responses in adult volunteers. PLoS Negl Trop Dis. 2018;12(4):e0006442. doi:10.1371/journal.pntd.0006442. - DOI - PMC - PubMed
-
- Jiang Z-D, Lowe B, Verenkar MP, Ashley D, Steffen R, Tornieporth N, Von Sonnenburg F, Waiyaki P, DuPont HL. Prevalence of enteric pathogens among international travelers with diarrhea acquired in Kenya (Mombasa), India (Goa), or Jamaica (Montego Bay). . The Journal of Infectious Diseases. 2002;185(4):497–502. doi:10.1086/338834. - DOI - PubMed
-
- Sack DA, Shimko J, Torres Bourgeois AL, Francia DS, Gustafsson B, Kärnell A, Nyquist I, Svennerholm AM. Randomised, double-blind, safety and efficacy of a killed oral vaccine for enterotoxigenic E. coli diarrhoea of travellers to Guatemala and Mexico. Vaccine. 2007;25:4392–4400. doi:10.1016/j.vaccine.2007.03.034. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
