PKA-RIIβ autophosphorylation modulates PKA activity and seizure phenotypes in mice
- PMID: 33649504
- PMCID: PMC7921646
- DOI: 10.1038/s42003-021-01748-4
PKA-RIIβ autophosphorylation modulates PKA activity and seizure phenotypes in mice
Abstract
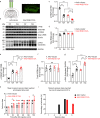
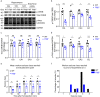
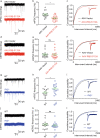
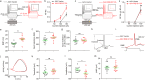
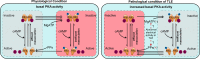
Temporal lobe epilepsy (TLE) is one of the most common and intractable neurological disorders in adults. Dysfunctional PKA signaling is causally linked to the TLE. However, the mechanism underlying PKA involves in epileptogenesis is still poorly understood. In the present study, we found the autophosphorylation level at serine 114 site (serine 112 site in mice) of PKA-RIIβ subunit was robustly decreased in the epileptic foci obtained from both surgical specimens of TLE patients and seizure model mice. The p-RIIβ level was negatively correlated with the activities of PKA. Notably, by using a P-site mutant that cannot be autophosphorylated and thus results in the released catalytic subunit to exert persistent phosphorylation, an increase in PKA activities through transduction with AAV-RIIβ-S112A in hippocampal DG granule cells decreased mIPSC frequency but not mEPSC, enhanced neuronal intrinsic excitability and seizure susceptibility. In contrast, a reduction of PKA activities by RIIβ knockout led to an increased mIPSC frequency, a reduction in neuronal excitability, and mice less prone to experimental seizure onset. Collectively, our data demonstrated that the autophosphorylation of RIIβ subunit plays a critical role in controlling neuronal and network excitabilities by regulating the activities of PKA, providing a potential therapeutic target for TLE.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

